
Acquired Hemophilia Treatment Market Report 2026
Global Outlook – By Treatment Approach (Bleeding Control Therapy (On-Demand Treatment), Inhibitor Eradication Therapy (Immunosuppressive Treatment)), By Route of Administration (Intravenous, Subcutaneous), By End User (Hospitals, Specialty Hematology Clinics, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Acquired Hemophilia Treatment Market Overview
• Acquired Hemophilia Treatment market size has reached to $12.83 billion in 2025 • Expected to grow to $15.91 billion in 2030 at a compound annual growth rate (CAGR) of 4.4% • Growth Driver: Growing Number Of Clinical Trials Fuels Acquired Hemophilia Treatment Market Growth • Market Trend: Advancements In Therapeutics Expand Effective Management Options In Acquired Hemophilia A • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Acquired Hemophilia Treatment Market?
Acquired hemophilia treatment refers to the therapeutic approaches used to manage and treat acquired hemophilia, a non-hereditary bleeding disorder that arises from a mutation or alteration in one of the genes responsible for producing clotting factor proteins essential for blood clot formation. The treatment involves strategies including controlling bleeding episodes and eradicating the inhibitors that target clotting factors. The main acquired hemophilia treatment includes on-demand and prophylaxis. On-demand treatment refers to the administration of clotting factor replacement therapy only in response to clinically significant bleeding. This treatment is prescribed for hemophilia A, hemophilia B, hemophilia C, and others by hospitals, clinics, and other end users.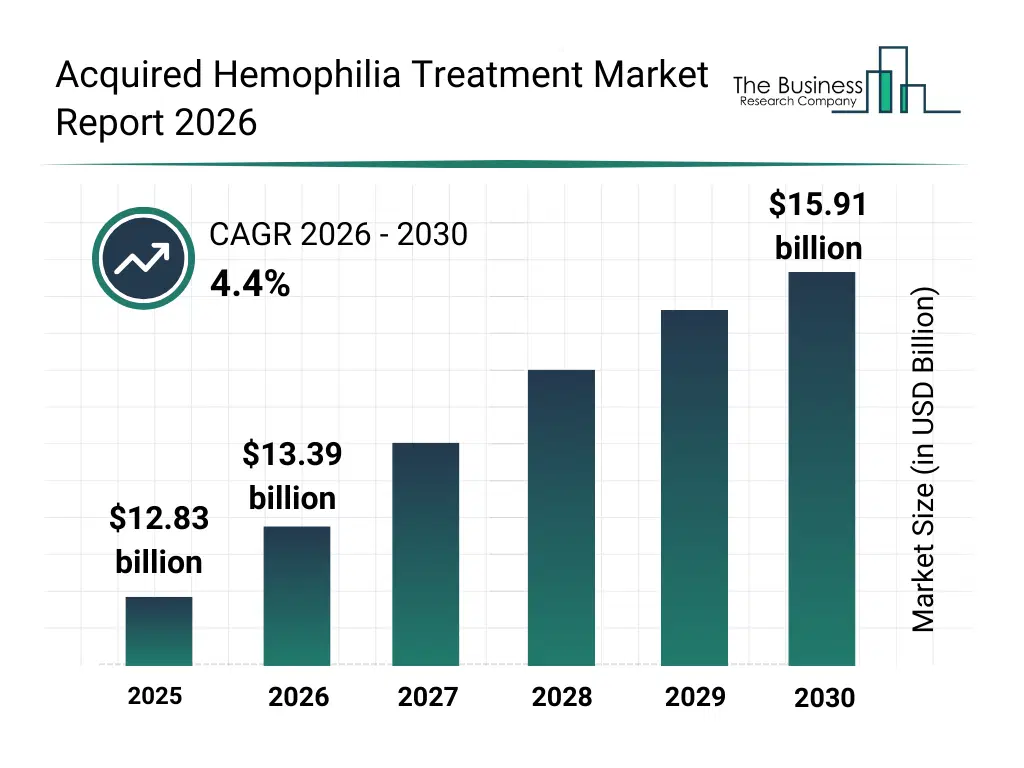
What Is The Acquired Hemophilia Treatment Market Size and Share 2026?
The acquired hemophilia treatment market size has grown steadily in recent years. It will grow from $12.83 billion in 2025 to $13.39 billion in 2026 at a compound annual growth rate (CAGR) of 4.4%. The growth in the historic period can be attributed to limited availability of specialized hemophilia treatments, rising prevalence of acquired hemophilia cases, dependence on hospital-based care, increasing awareness of bleeding disorders, adoption of on-demand treatment protocols.What Is The Acquired Hemophilia Treatment Market Growth Forecast?
The acquired hemophilia treatment market size is expected to see steady growth in the next few years. It will grow to $15.91 billion in 2030 at a compound annual growth rate (CAGR) of 4.4%. The growth in the forecast period can be attributed to technological advancements in gene therapy, expansion of prophylaxis treatment adoption, growth of personalized medicine in hematology, rising investment in hemophilia research, increasing access to advanced treatment options in emerging regions. Major trends in the forecast period include rising adoption of clotting factor replacement therapies, increasing use of gene therapy for hemophilia prophylaxis, expansion of personalized treatment plans for acquired hemophilia, growing awareness and early diagnosis programs, focus on reducing bleeding episodes and inhibitor eradication.Global Acquired Hemophilia Treatment Market Segmentation
1) By Treatment Approach: Bleeding Control Therapy (On-Demand Treatment), Inhibitor Eradication Therapy (Immunosuppressive Treatment) 2) By Route of Administration: Intravenous, Subcutaneous 3) By End User: Hospitals, Specialty Hematology Clinics, Other End Users Subsegments: 1) By Bleeding Control Therapy (On-Demand Treatment): Bypassing Agents, Recombinant Activated Factor VII (rFVIIa), Activated Prothrombin Complex Concentrates (aPCC), Recombinant Porcine Factor VIII, Desmopressin 2) By Inhibitor Eradication Therapy (Immunosuppressive Treatment): Corticosteroid Monotherapy, Corticosteroids + Cytotoxic Agents (e.g., Cyclophosphamide), Rituximab-Based Immunotherapy, Combination Immunosuppressive RegimensWhat Is The Driver Of The Acquired Hemophilia Treatment Market?
A growing number of ongoing clinical trials is expected to propel the growth of the acquired hemophilia treatment market going forward. Clinical trials are research studies conducted with human participants to evaluate the safety and effectiveness of new medical treatments, interventions, or diagnostic procedures. Clinical trials assess the safety and efficacy of new treatments for acquired hemophilia, advancing research to improve patient outcomes and establish effective therapeutic interventions. For instance, in December 2024, according to the Association of the British Pharmaceutical Industry, a UK-based trade association, the count of pharmaceutical industry trials launched in the UK increased for the second consecutive year, rising from 411 in 2022 to 426 in 2023. Therefore, a growing number of ongoing clinical trials is driving the growth of the acquired hemophilia treatment industry.Key Players In The Global Acquired Hemophilia Treatment Market
Major companies operating in the acquired hemophilia treatment market are Roche Holding AG, Novo Nordisk A/S, Takeda Pharmaceutical Company Limited, Pfizer Inc., Bayer AG, CSL Limited, Grifols SA, Octapharma AG, Kedrion Biopharma Inc., Sanofi SA, Sobi Inc., Chugai Pharmaceutical Co. Ltd., Novartis AG, Bristol-Myers Squibb Company, Johnson & Johnson, Medexus Pharmaceuticals Inc., Ferring Pharmaceuticals BV, Hema Biologics LLC, Bio Products Laboratory Ltd., LFB SAGlobal Acquired Hemophilia Treatment Market Trends and Insights
Major companies operating in the acquired hemophilia treatment market are focused on providing drugs with additional measures and getting them approved to sustain their position in the market. Drug approvals to include routine prophylaxis are increasing, mainly due to the need for more effective and safer. For instance, in June 2023, Chugai Pharmaceutical Co., Ltd., a Japan-based drug manufacturer, presented final analysis data from its Japanese Phase III clinical trial (AGEHA Study) on Hemlibra (emicizumab) for acquired hemophilia A at the 31st ISTH Annual Congress. The study included patients receiving immunosuppressive therapy as well as those ineligible for such therapy. Hemlibra demonstrated long-term efficacy in managing bleeding events, supporting its expanded use in individuals with acquired hemophilia A who require prompt and effective treatment.What Are Latest Mergers And Acquisitions In The Acquired Hemophilia Treatment Market?
In June 2023, Baudax Bio., a US-based pharmaceutical company, acquired TeraImmune Inc. for an undisclosed amount. With this acquisition, Baudax Bio aimed to combine the top-tier scientific expertise of the TeraImmune team with the Baudax team's established proficiency in executing clinical development programs and incorporate TeraImmune's TI-168 asset into Baudax's portfolio. This asset was a promising next-generation, autologous FVIII TCR-Treg cell therapy candidate designed to neutralize clotting factor VIII (FVIII) inhibitors in Hemophilia A patients. TeraImmune Inc. is a US-based advanced research company developing treatments for hemophilia.Regional Outlook
North America was the largest region in the acquired hemophilia treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Acquired Hemophilia Treatment Market?
The acquired hemophilia treatment market consists of revenues earned by entities by providing services such as eradication of inhibitors, individualized therapeutic approach, multidisciplinary approach, and prevention services. The market value includes the value of related goods sold by the service provider or included within the service offering. The acquired hemophilia treatment market also includes sales of immunosuppressive agents including cyclosporine A, and azathioprine. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Acquired Hemophilia Treatment Market Report 2026?
The acquired hemophilia treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the acquired hemophilia treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Acquired Hemophilia Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $13.39 billion |
| Revenue Forecast In 2035 | $15.91 billion |
| Growth Rate | CAGR of 4.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Approach, Route of Administration, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Roche Holding AG, Novo Nordisk A/S, Takeda Pharmaceutical Company Limited, Pfizer Inc., Bayer AG, CSL Limited, Grifols SA, Octapharma AG, Kedrion Biopharma Inc., Sanofi SA, Sobi Inc., Chugai Pharmaceutical Co. Ltd., Novartis AG, Bristol-Myers Squibb Company, Johnson & Johnson, Medexus Pharmaceuticals Inc., Ferring Pharmaceuticals BV, Hema Biologics LLC, Bio Products Laboratory Ltd., LFB SA |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
