
Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Report 2026
Global Outlook – By Product (Gene Therapy, Cell Therapy, Tissue Engineered), By Phase (Phase I, Phase II, Phase III, Phase IV), By Indication (Oncology, Cardiology, Central Nervous System And Musculoskeletal, Infectious Disease, Dermatology, Endocrine, Metabolic, Genetic, Immunology And Inflammation, Ophthalmology, Hematology) - Market Size, Trends, And Global Forecast 2026-2035
Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Overview
• Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) market size has reached to $6.2 billion in 2025 • Expected to grow to $12.55 billion in 2030 at a compound annual growth rate (CAGR) of 14.8% • Growth Driver: Rising Clinical Trial Numbers Fuel Growth In The ATMP CDMO Market • Market Trend: Transforming Cell Therapy Manufacturing With Innovations In Advanced Therapy Medicinal Products • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market?
An advanced therapy medicinal product contract development and manufacturing organization (CDMO) is a specialized service provider that assists in the development and manufacturing of advanced therapy medicinal products. The objective of an advanced therapy medicinal product CDMO is to provide the expertise, infrastructure, and resources required for advanced therapy medicinal product development and manufacturing. The main types of products of advanced therapy medicinal products contract development and manufacturing organization (CDMO) include gene therapy, cell therapy, and tissue engineered. Gene therapy involves the introduction, removal, or alteration of genetic material within a patient's cells to treat or prevent disease. This can be done using viral or non-viral vectors to deliver therapeutic genes to target cells in several phases, such as phase I, phase II, phase III, and phase IV for the treatment of various indications, including oncology, cardiology, central nervous system and musculoskeletal, infectious disease, dermatology, endocrine, metabolic, genetic, immunology and inflammation, ophthalmology, and hematology.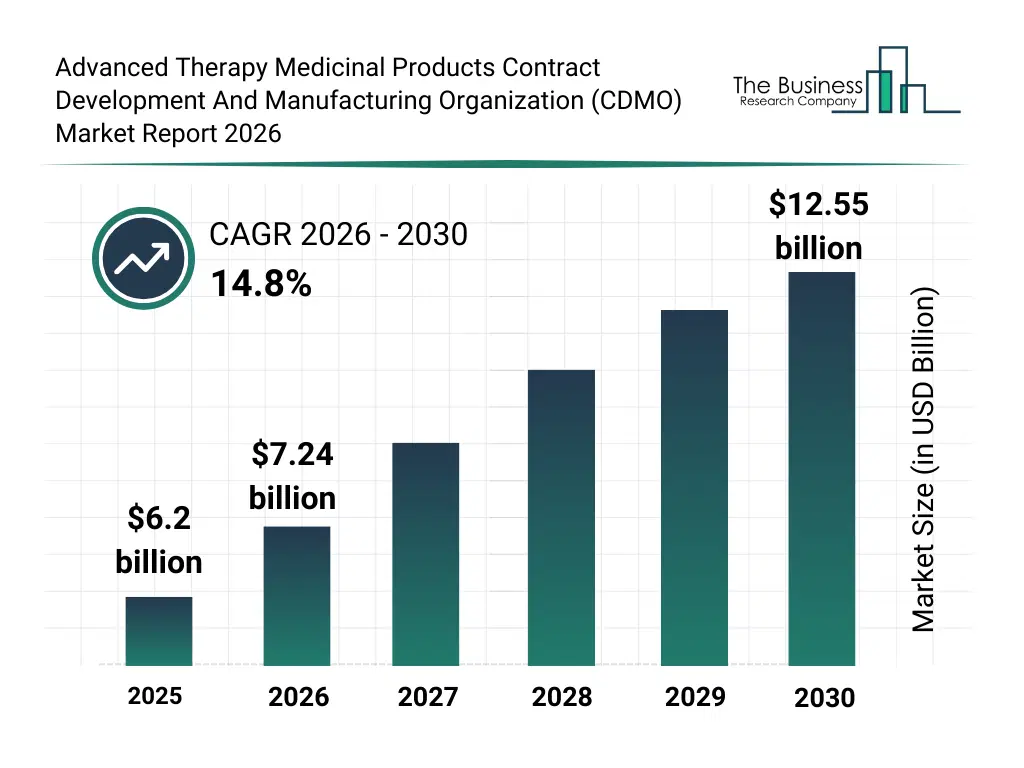
What Is The Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Size and Share 2026?
The advanced therapy medicinal products contract development and manufacturing organization (cdmo) market size has grown rapidly in recent years. It will grow from $6.2 billion in 2025 to $7.24 billion in 2026 at a compound annual growth rate (CAGR) of 16.7%. The growth in the historic period can be attributed to limited availability of specialized cdmo services, increasing r&d investment in advanced therapies, early adoption of gene and cell therapies, rising prevalence of complex diseases, growth of small biotech startups.What Is The Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Growth Forecast?
The advanced therapy medicinal products contract development and manufacturing organization (cdmo) market size is expected to see rapid growth in the next few years. It will grow to $12.55 billion in 2030 at a compound annual growth rate (CAGR) of 14.8%. The growth in the forecast period can be attributed to advancements in scalable manufacturing technologies, increasing collaborations between cdmos and pharma, growth of autologous and allogeneic therapies, rising global demand for personalized medicine, expansion into emerging markets. Major trends in the forecast period include rising demand for cdmo services in advanced therapy development, expansion of gene, cell, and tissue engineered therapies, increasing focus on regulatory compliance and quality standards, growth in outsourced manufacturing for biopharmaceutical companies, adoption of scalable and flexible manufacturing platforms.Global Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Segmentation
1) By Product: Gene Therapy, Cell Therapy, Tissue Engineered 2) By Phase: Phase I, Phase II, Phase III, Phase IV 3) By Indication: Oncology, Cardiology, Central Nervous System And Musculoskeletal, Infectious Disease, Dermatology, Endocrine, Metabolic, Genetic, Immunology And Inflammation, Ophthalmology, Hematology Subsegments: 1) By Gene Therapy: Viral Vectors, Non-Viral Vectors, Dna Or Rna-Based Products 2) By Cell Therapy: Autologous Cell Therapy, Allogeneic Cell Therapy, Stem Cell Therapy 3) By Tissue Engineered: Skin Substitutes, Cartilage And Bone Tissue, Vascular Grafts And OrgansWhat Is The Driver Of The Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market?
The increasing number of clinical trials is expected to propel the growth of the advanced therapy medicinal products CDMO market going forward. Clinical trials are research studies conducted in humans to evaluate the safety and efficacy of medical interventions such as drugs, treatments, devices, or preventive measures. Clinical trial numbers are rising to meet the increasing demand for innovative treatments and to address a growing global disease burden. With the use of advanced therapy medicinal products, the demand for CDMO manufacturing services rises, leveraging specialized expertise and infrastructure. Clinical trial data aids in optimizing manufacturing, ensuring scalability, and quality maintenance to meet larger clinical and commercial needs. For instance, in July 2024, according to the Royal Pharmaceutical Society, a UK-based professional membership body, the number of people recruited to clinical trials in the UK in 2023/2024 was 349,019, an increase from 342,390 people in 2022/2023. Therefore, the increasing number of clinical trials is driving the growth of the advanced therapy medicinal products CDMO market.Key Players In The Global Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market
Major companies operating in the advanced therapy medicinal products contract development and manufacturing organization (cdmo) market are Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Catalent Inc., Charles River Laboratories International Inc., Recipharm AB, KBI Biopharma, Takara Bio Inc., Rentschler Biopharma SE, Ajinomoto Co. Inc., ElevateBio LLC, Fujifilm Diosynth Biotechnologies, Axplora Group GmbH, WuXi Advanced Therapies Inc., Celonic Group, Batavia Biosciences B.V., BioCentriq, Cytovance Biologics, Andelyn Biosciences, Genezen, AGC Biologics, ABL Manufacturing, Minaris Regenerative Medicine, Porton Advanced SolutionsGlobal Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Trends and Insights
Major companies operating in the advanced therapy medicinal products CDMO market are developing advanced cell therapy manufacturing services to overcome critical issues in cell therapy manufacturing and speed the development activities of their partners. Cell therapy manufacturing services are contract development and manufacturing organization services that offer services which vary from early preclinical development to late-stage clinical trials and commercialization. For instance, in January 2024, Pluristem Therapeutics Inc., an Israel-based biotech company that transforms cells into solutions, launched PluriCDMO, a new business division offering cell therapy manufacturing. It utilizes a patented bioreactor system that allows for 3D cell multiplication, facilitating the production of a variety of cell types such as stem cells, induced pluripotent stem cells, exosomes, and immune therapies. PluriCDMO desires to provide high-quality, vital medicines to patients while maintaining unique batch-to-batch consistency in a scalable and cost-effective manner in good manufacturing practice settings.What Are Latest Mergers And Acquisitions In The Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market?
In January 2025, Altaris, LLC, a US-based investment firm focused on healthcare and building companies in the medical-product and biotech services space, acquired Minaris Regenerative Medicine from Resonac Corporation for an undisclosed amount. Through this acquisition, Altaris aims to deepen its footprint in the advanced therapy medicinal products (ATMP) contract development and manufacturing (CDMO) sector and accelerate its strategy to scale life-changing cell and gene therapies globally. Minaris Regenerative Medicine is a US-based CDMO providing autologous and allogeneic cell therapy manufacturing services across clinical and commercial stages.Regional Outlook
North America was the largest region in the advanced therapy medicinal product contract development and manufacturing organization (CDMO) market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market?
The advanced therapy medicinal product contract development and manufacturing organization (CDMO) market includes revenues earned by entities by providing services such as process development, analytical method development, good manufacturing practice manufacturing, and quality control testing. The market value includes the value of related goods sold by the service provider or included within the service offering. The advanced therapy medicinal products CDMO market also includes sales of bioreactors and fermenters, cryopreservation equipment, and laboratory instruments. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Report 2026?
The advanced therapy medicinal products contract development and manufacturing organization (cdmo) market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the advanced therapy medicinal products contract development and manufacturing organization (cdmo) industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Advanced Therapy Medicinal Products Contract Development And Manufacturing Organization (CDMO) Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.24 billion |
| Revenue Forecast In 2035 | $12.55 billion |
| Growth Rate | CAGR of 16.7% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Phase, Indication |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Lonza Group AG, Catalent Inc., Charles River Laboratories International Inc., Recipharm AB, KBI Biopharma, Takara Bio Inc., Rentschler Biopharma SE, Ajinomoto Co. Inc., ElevateBio LLC, Fujifilm Diosynth Biotechnologies, Axplora Group GmbH, WuXi Advanced Therapies Inc., Celonic Group, Batavia Biosciences B.V., BioCentriq, Cytovance Biologics, Andelyn Biosciences, Genezen, AGC Biologics, ABL Manufacturing, Minaris Regenerative Medicine, Porton Advanced Solutions |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
