
Clinical Oncology Next Generation Sequencing Market Report 2026
Global Outlook – By Technology (Ion Semiconductor Sequencing, Pyro-Sequencing, Synthesis Sequencing, Real Time Sequencing, Ligation Sequencing, Reversible Dye Termination Sequencing, Nano-Pore Sequencing), By Application (Screening, Companion Diagnostics, Other Diagnostics), By End User (Hospital Laboratories, Clinical Research Organizations, Diagnostic Laboratories) – Market Size, Trends, Strategies, and Forecast to 2035
Clinical Oncology Next Generation Sequencing Market Overview
• Clinical Oncology Next Generation Sequencing market size has reached to $0.58 billion in 2025 • Expected to grow to $1.04 billion in 2030 at a compound annual growth rate (CAGR) of 12.1% • Growth Driver: Rising Prevalence Of Cancer Driving Market Growth Due To Increased Demand For Precision Oncology Solutions • Market Trend: Development Of Lightspeed Technology For Ultra-Fast, Low-Cost Somatic Variant Analysis • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Clinical Oncology Next Generation Sequencing Market?
Next generation sequencing (NGS) is a process used to determine the sequence of nucleotides in a section of DNA and is used in oncology research. NGS is used to identify rare cancer mutations and familial cancer mutation carriers and to provide molecular rationale for appropriate targeted therapy. The main technologies in clinical oncology next-generation sequencing are ion semiconductor sequencing, pyro-sequencing, synthesis sequencing, real-time sequencing, ligation sequencing, reversible dye termination sequencing, and nanopore sequencing. Ion semiconductor sequencing is a technique of DNA sequencing found on the observation of hydrogen ions that are released throughout the polymerization of DNA. The clinical oncology next-generation sequencing is used in various applications such as screening, companion diagnostics, and other diagnostics that are used by various end-users such as hospital laboratories, clinical research organizations, and diagnostic laboratories.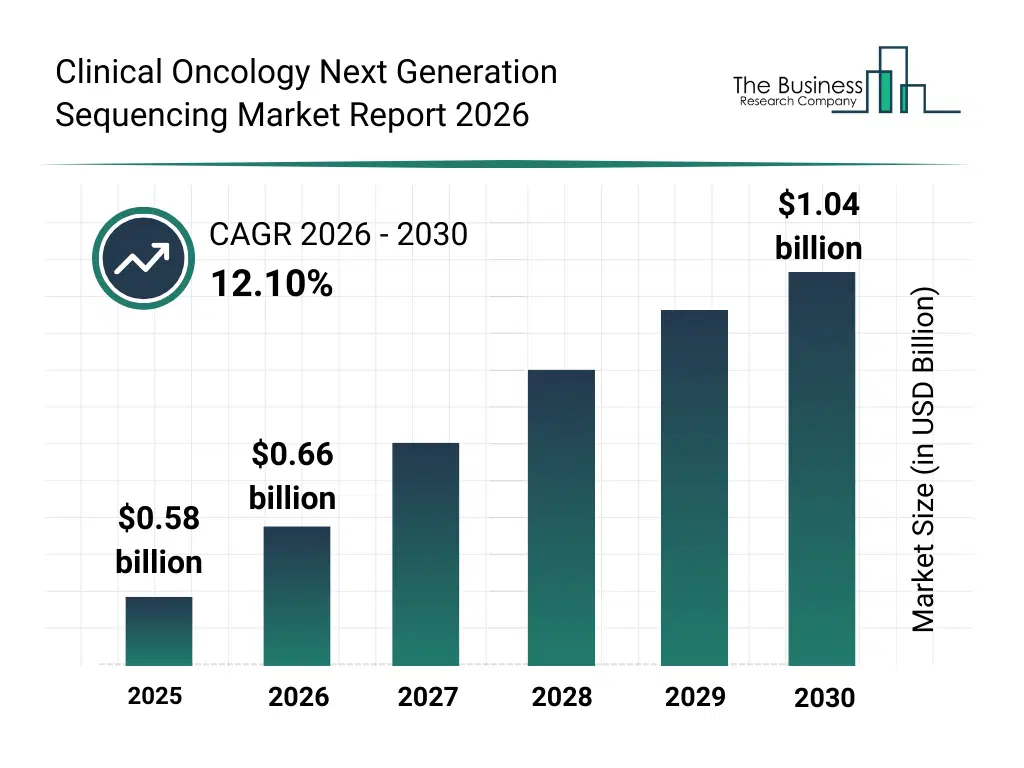
What Is The Clinical Oncology Next Generation Sequencing Market Size and Share 2026?
The clinical oncology next generation sequencing market size has grown rapidly in recent years. It will grow from $0.58 billion in 2025 to $0.66 billion in 2026 at a compound annual growth rate (CAGR) of 13.3%. The growth in the historic period can be attributed to expansion of cancer genomics research, increased availability of sequencing platforms, growth in oncology clinical trials, rising adoption of molecular diagnostics, declining sequencing costs.What Is The Clinical Oncology Next Generation Sequencing Market Growth Forecast?
The clinical oncology next generation sequencing market size is expected to see rapid growth in the next few years. It will grow to $1.04 billion in 2030 at a compound annual growth rate (CAGR) of 12.1%. The growth in the forecast period can be attributed to increasing demand for precision oncology, rising investments in genomic medicine, expansion of hospital-based sequencing laboratories, growing use of targeted cancer therapies, continuous advancements in sequencing technologies. Major trends in the forecast period include increasing adoption of ngs-based oncology diagnostics, rising use of companion diagnostic applications, growing integration of bioinformatics and data analytics, expansion of personalized cancer treatment approaches, enhanced focus on rare mutation detection.Global Clinical Oncology Next Generation Sequencing Market Segmentation
1) By Technology: Ion Semiconductor Sequencing, Pyro-Sequencing, Synthesis Sequencing, Real Time Sequencing, Ligation Sequencing, Reversible Dye Termination Sequencing, Nano-Pore Sequencing 2) By Application: Screening, Companion Diagnostics, Other Diagnostics 3) By End User: Hospital Laboratories, Clinical Research Organizations, Diagnostic Laboratories Subsegments: 1) By Ion Semiconductor Sequencing: Ion Proton System, Ion PGM System, Ion S5 System, Other Ion Semiconductor Sequencing Technologies 2) By Pyro-Sequencing: 454 Pyrosequencing, Other Pyro-Sequencing Technologies 3) By Synthesis Sequencing: Illumina Sequencing Technology, SOLiD Sequencing Technology, Other Synthesis Sequencing Technologies 4) By Real-Time Sequencing: PacBio RS II System, Sequel System, Other Real-Time Sequencing Technologies 5) By Ligation Sequencing: Helicos Single Molecule Sequencing, Other Ligation Sequencing Technologies 6) By Reversible Dye Termination Sequencing: Illumina HiSeq, Illumina NextSeq, Other Reversible Dye Termination Sequencing Technologies 7) By Nano-Pore Sequencing: Oxford Nanopore Technologies (MinION, GridION, PromethION), Other Nano-Pore Sequencing TechnologiesWhat Is The Driver Of The Clinical Oncology Next Generation Sequencing Market?
The rising prevalence of cancer is expected to propel the growth of the clinical oncology next-generation sequencing market going forward. Cancer is a term used to describe a group of diseases characterized by the uncontrolled growth and spread of abnormal cells in the body. The rising prevalence of cancer is primarily due to population aging, as cancer risk significantly increases with age. NGS is used to identify new and rare mutations in cancer, providing a deeper understanding of the molecular underpinnings of individual tumors. For instance, in February 2024, according to the World Health Organization, a Switzerland-based-based public health agency of United Nations, over 35 million new cancer cases are projected for 2050, reflecting a 77% increase compared with the estimated 20 million cases reported in 2022. Therefore, the rising prevalence of cancer is driving the growth of the clinical oncology next-generation sequencing market.Key Players In The Global Clinical Oncology Next Generation Sequencing Market
Major companies operating in the clinical oncology next generation sequencing market are Thermo Fisher Scientific, Oxford Nanopore Technologies Ltd., QIAGEN N.V., Myriad Genetics Inc., Illumina Inc., F. Hoffmann-La Roche Ltd., PerkinElmer Inc., Agilent Technologies Inc., Pacific Biosciences of California Inc., Caris Life Sciences, Paradigm Diagnostics, GATC Biotech AG, Macrogen Inc., DNASTAR Inc., Exosome Diagnostics Inc., Biomatters Ltd., Partek Inc., Foundation Medicine Inc., Becton Dickinson and Company (BD), Takara Bio Inc., Knome Inc., Genomatix Software GmbH, CLC bio, GnuBIO Inc., Bio-Rad Laboratories Inc., BGI Genomics Co. Ltd., Guardant Health Inc., Invitae Corporation, Natera Inc., NeoGenomics Laboratories Inc., Sysmex Corporation, Veracyte Inc., Zymo Research Corporation, ArcherDX Inc., Karius Inc., OncoDNA S.A., Personal Genome Diagnostics Inc., PierianDx Inc.Global Clinical Oncology Next Generation Sequencing Market Trends and Insights
Major companies operating in the clinical oncology next-generation sequencing (NGS) market are focusing on technological development, such as embedding software accelerators that drastically reduce the time and cost of second-level NGS analysis, enabling labs to deliver results faster while shrinking computational resource needs. These accelerator technologies support comprehensive genomic profiling and make high-throughput cancer panels more accessible to both large centralized laboratories and decentralized diagnostic settings. For example, in January 2024, QIAGEN Digital Insights, a Germany-based bioinformatics company, launched an enhanced version of its CLC Genomics Workbench Premium featuring LightSpeed technology, now supporting somatic cancer secondary analysis. The LightSpeed accelerator can analyze a 275-gene cancer panel at 3377× coverage in just 6 minutes, costing less than US$ 0.72 per test in standard cloud environments. LightSpeed is optimized to deliver high speed, accuracy, reduced power consumption, and no need for new hardware or additional licenses beyond the existing CLC platform.What Are Latest Mergers And Acquisitions In The Clinical Oncology Next Generation Sequencing Market?
In January 2023, Agilent Technologies, a US-based scientific equipment manufacturer, acquired Avida Biomed for an undisclosed amount. Through this acquisition, Agilent Technologies aims to enhance its high-performance target enrichment workflows for next-generation sequencing applications, particularly in cancer research, by integrating Avida's innovative technologies to improve precision medicine approaches and expanding its SureSelect portfolio into the clinical research and diagnostics markets. Avida Biomed, a US-based biotechnology company, offers clinical oncology next-generation sequencing.Regional Outlook
North America was the largest region in the clinical oncology next-generation sequencing market in 2025. Asia-Pacific was the second largest region in the clinical oncology next-generation sequencing market. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Clinical Oncology Next Generation Sequencing Market?
The clinical oncology next-generation sequencing market consists of sales of genetic sequencing equipment with advanced features and technologies such as ion semiconductors sequencing, pyro-sequencing, synthesis sequencing (SBS), real-time sequencing (SMRT), ligation sequencing, and other techniques that are used in oncology research for determining the nucleotide sequence leading to clinical oncology. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Clinical Oncology Next Generation Sequencing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.66 billion |
| Revenue Forecast In 2035 | $1.04 billion |
| Growth Rate | CAGR of 13.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Thermo Fisher Scientific, Oxford Nanopore Technologies Ltd., QIAGEN N.V., Myriad Genetics Inc., Illumina Inc., F. Hoffmann-La Roche Ltd., PerkinElmer Inc., Agilent Technologies Inc., Pacific Biosciences of California Inc., Caris Life Sciences, Paradigm Diagnostics, GATC Biotech AG, Macrogen Inc., DNASTAR Inc., Exosome Diagnostics Inc., Biomatters Ltd., Partek Inc., Foundation Medicine Inc., Becton Dickinson and Company (BD), Takara Bio Inc., Knome Inc., Genomatix Software GmbH, CLC bio, GnuBIO Inc., Bio-Rad Laboratories Inc., BGI Genomics Co. Ltd., Guardant Health Inc., Invitae Corporation, Natera Inc., NeoGenomics Laboratories Inc., Sysmex Corporation, Veracyte Inc., Zymo Research Corporation, ArcherDX Inc., Karius Inc., OncoDNA S.A., Personal Genome Diagnostics Inc., PierianDx Inc. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
