
COVID-19 Drug Associated APIs Market Report 2026
Global Outlook – By Drug Class (Antimalarials, Bronchodilators, Antibiotics, Antivirals, Other Drug Class), By Synthesis Type (Synthetic, Biotech), By Business Mode (Captive API, Merchant API) – Market Size, Trends, Strategies, and Forecast to 2035
COVID-19 Drug Associated APIs Market Overview
• COVID-19 Drug Associated APIs market size has reached to $7.14 billion in 2025 • Expected to grow to $9.69 billion in 2030 at a compound annual growth rate (CAGR) of 6.4% • Growth Driver: Government Initiatives Catalysts For Growth In The COVID-19 Drug-Associated API Market • Market Trend: Innovative Strategies In The COVID-19 Drug-Associated APIs Market Focus On Oral Treatments • North America was the largest region in 2025 and Middle East is the fastest growing region.What Is Covered Under COVID-19 Drug Associated APIs Market?
COVID-19 drug-associated APIs refer to the active pharmaceutical components that are employed in the production of COVID-19 medications. Chemicals called active pharmaceutical components serve as the foundation for pharmaceutical drugs and medicines and are responsible for pharmacological activity in the human body. The main classes of drugs in COVID-19 drug-associated APIs are antimalarials, bronchodilators, antibiotics, antivirals, and others. Antimalarial drugs, or simply antimalarials, are antiparasitic chemical agents that can be used to cure or prevent malaria. They are generally organically sourced. The different types of drugs include generic, and branded, and involve various modes of business such as captive API and merchant API.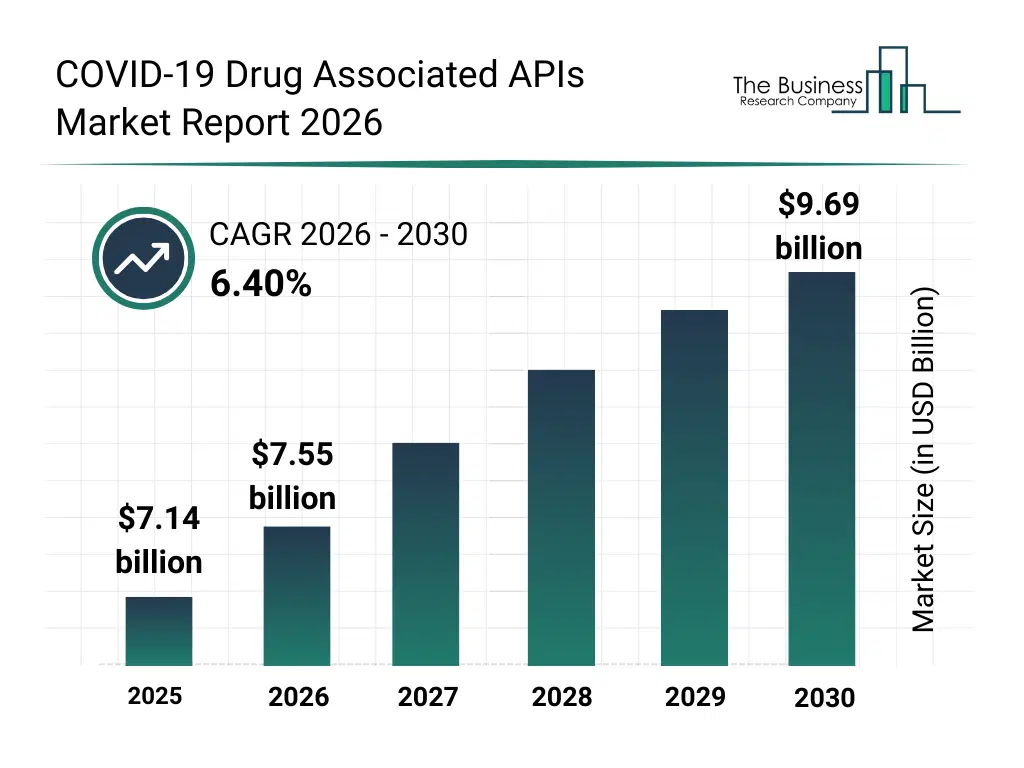
What Is The COVID-19 Drug Associated APIs Market Size and Share 2026?
The covid-19 drug associated apis market size has grown strongly in recent years. It will grow from $7.14 billion in 2025 to $7.55 billion in 2026 at a compound annual growth rate (CAGR) of 5.8%. The growth in the historic period can be attributed to initial surge in covid-19 cases, urgent need for repurposed drugs, limited availability of antiviral apis, dependency on imports from asia-pacific, accelerated emergency approvals.What Is The COVID-19 Drug Associated APIs Market Growth Forecast?
The covid-19 drug associated apis market size is expected to see strong growth in the next few years. It will grow to $9.69 billion in 2030 at a compound annual growth rate (CAGR) of 6.4%. The growth in the forecast period can be attributed to development of next-generation antiviral and immunomodulator apis, expansion of domestic api manufacturing, increasing adoption of biotech synthesis, rising government funding for pandemic preparedness, growing demand for diversified covid-19 treatment options. Major trends in the forecast period include rising demand for antiviral and antimalarial apis, increased focus on captive and merchant api manufacturing, expansion of synthetic and biotech-based api production, growing investment in research and development for covid-19 drugs, enhanced regulatory oversight on api quality and supply chains.Global COVID-19 Drug Associated APIs Market Segmentation
1) By Drug Class: Antimalarials, Bronchodilators, Antibiotics, Antivirals, Other Drug Class 2) By Synthesis Type: Synthetic, Biotech 3) By Business Mode: Captive API, Merchant API Subsegments: 1) By Antimalarials: Hydroxychloroquine, Chloroquine 2) By Bronchodilators: Beta-Agonists, Anticholinergics 3) By Antibiotics: Macrolides, Tetracyclines 4) By Antivirals: Remdesivir, Favipiravir, Lopinavir Or Ritonavir 5) By Other Drug Classes: Corticosteroids, ImmunomodulatorsWhat Is The Driver Of The COVID-19 Drug Associated APIs Market?
Government initiatives for research and development in healthcare are expected to propel the growth of the COVID-19 drug-associated API market going forward. Government initiatives refer to specific actions, programs, policies, or projects undertaken by government authorities at various levels (local, regional, national, or international) to address specific issues, achieve certain goals, or create positive changes in societyGovernment initiatives for research and development in healthcare are rising to promote innovation and accelerate the discovery of new treatments that address critical public health challenges. COVID-19 drug-associated APIs help government initiatives for research and development in healthcare by providing essential active ingredients that support rapid development, testing, and production of treatments and vaccines. For instance, in April 2025, according to the Office for National Statistics (ONS), a UK-government agency, UK government net R&D expenditure was £17.4 ($20.10) billion in 2023, up from £16.1 billion in 2022. Therefore, government initiatives for research and development in healthcare are driving the growth of the COVID-19 drug-associated API market.Key Players In The Global COVID-19 Drug Associated APIs Market
Major companies operating in the covid-19 drug associated apis market are Dr. Reddy's Laboratories Ltd., Lianyungang Guike Pharmaceutical CO. LTD., Alembic Pharmaceuticals Ltd., Wockhardt Ltd., Sandoz Srl, Lupin Limited, Aurobindo Pharma Ltd., Shanghai Shyndec Pharmaceutical Co. Ltd., Zhejiang Yatai Pharmaceutical Co. Ltd., Zhejiang Guobang Pharmaceutical Co Ltd., Cipla Limited, Shandong Boyuan Pharmaceutical Co. Ltd., Aspiro Pharma Ltd., Qilu Antibiotics Pharmaceutical Co. Ltd., Shenzhen China Resources Jiuxin Pharmaceutical Co. Ltd., Zhejiang Cheng Yi Pharmaceutical, Star Lake Bioscience Co.Inc., Hetero Labs Ltd., Geno Pharmaceuticals Ltd., Mylan Laboratories Ltd., Ipca Laboratories Limited, Zydus Takeda Healthcare Private Limited, Mangalam Drugs & Organics, Wallace Pharmaceuticals, Zhejiang Hisun Pharmaceutical Co. Ltd., Zhejiang Huahai Pharmaceutical Co. Ltd., Zhejiang Jiuzhou Pharmaceutical Co. Ltd., Zhejiang NHU Co. Ltd., Zhejiang Xianju Pharmaceutical Co. Ltd., Zhejiang Yongtai Technology Co. Ltd., Zhejiang Zhebei Pharmaceutical Co. Ltd., Zhejiang Tianyu Pharmaceutical Co. Ltd., Zhejiang Aisheng Pharmaceutical Co. Ltd., Zhejiang Aokang Pharmaceutical Co. Ltd.Global COVID-19 Drug Associated APIs Market Trends and Insights
Major companies operating in the COVID-19 drug-associated APIs market are increasing their focus on introducing innovative products, such as oral treatment for COVID-19, to gain a competitive edge in the market. Oral treatment for COVID-19 refers to medications that are taken by mouth (orally) to manage and treat the infection caused by the SARS-CoV-2 virus, which is responsible for COVID-19. For instance, in May 2023, Pfizer Inc., a US-based pharmaceutical company, launched PAXLOVID, which was approved by the FDA. The oral investigational drug, a SARS-CoV-2 protease inhibitor, has shown an 89% reduction in hospitalization or death risk compared to a placebo in high-risk, non-hospitalized adults with COVID-19. Designed for early administration, it aims to be prescribed at the first sign of infection or exposure awareness, potentially preventing severe illness, hospitalization, and death.What Are Latest Mergers And Acquisitions In The COVID-19 Drug Associated APIs Market?
In January 2023, Sterling Pharma Solutions, a US-based pharmaceutical company, acquired an active pharmaceutical ingredient (API) manufacturing facility from Novartis for an undisclosed sum. With this acquisition, Sterling's expanding API manufacturing capabilities, as well as the deal's ongoing supply arrangement with Novartis, are going to keep manufacturing several APIs for cardiovascular, immunology, and oncology medications. Novartis AG is a US-based pharmaceutical company that manufactures active pharmaceutical ingredients (API).Regional Insights
North America was the largest region in the COVID-19 drug-associated APIs market in 2025. Middle East is expected to be the fastest growing region in the global COVID-19 drug associated APIs market share during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the COVID-19 Drug Associated APIs Market?
The COVID-19 drug-associated APIs market consists of sales of acetaminophen (Tylenol) and ibuprofen (Motrin, Advil). Values in this market are factory gate values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the COVID-19 Drug Associated APIs Market Report 2026?
The covid-19 drug associated apis market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the covid-19 drug associated apis industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.COVID-19 Drug Associated APIs Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.55 billion |
| Revenue Forecast In 2035 | $9.69 billion |
| Growth Rate | CAGR of 5.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug Class, Synthesis Type, Business Mode |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Dr. Reddy's Laboratories Ltd., Lianyungang Guike Pharmaceutical CO. LTD., Alembic Pharmaceuticals Ltd., Wockhardt Ltd., Sandoz Srl, Lupin Limited, Aurobindo Pharma Ltd., Shanghai Shyndec Pharmaceutical Co. Ltd., Zhejiang Yatai Pharmaceutical Co. Ltd., Zhejiang Guobang Pharmaceutical Co Ltd., Cipla Limited, Shandong Boyuan Pharmaceutical Co. Ltd., Aspiro Pharma Ltd., Qilu Antibiotics Pharmaceutical Co. Ltd., Shenzhen China Resources Jiuxin Pharmaceutical Co. Ltd., Zhejiang Cheng Yi Pharmaceutical, Star Lake Bioscience Co.Inc., Hetero Labs Ltd., Geno Pharmaceuticals Ltd., Mylan Laboratories Ltd., Ipca Laboratories Limited, Zydus Takeda Healthcare Private Limited, Mangalam Drugs & Organics, Wallace Pharmaceuticals, Zhejiang Hisun Pharmaceutical Co. Ltd., Zhejiang Huahai Pharmaceutical Co. Ltd., Zhejiang Jiuzhou Pharmaceutical Co. Ltd., Zhejiang NHU Co. Ltd., Zhejiang Xianju Pharmaceutical Co. Ltd., Zhejiang Yongtai Technology Co. Ltd., Zhejiang Zhebei Pharmaceutical Co. Ltd., Zhejiang Tianyu Pharmaceutical Co. Ltd., Zhejiang Aisheng Pharmaceutical Co. Ltd., Zhejiang Aokang Pharmaceutical Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
