
Duchenne Muscular Dystrophy Market Report 2026
Global Outlook – By Therapy (Mutation Suppression, Exon Skipping Approach), By Therapeutic Class (Molecular Based Therapies, Steroid Therapy), By Route Of Administration (Oral, Parenteral, Other Routes Of Administration), By Distribution Channel (Hospital Pharmacy, Online Pharmacy, Retail Pharmacy), By End User (Hospitals, Homecare, Specialty Clinics, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Duchenne Muscular Dystrophy Market Overview
• Duchenne Muscular Dystrophy market size has reached to $1.25 billion in 2025 • Expected to grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 8.5% • Growth Driver: Rising Prevalence Of Chronic Diseases Fuels Growth In The Duchenne Muscular Dystrophy Market • Market Trend: Technological Advancements In Clinical Trial Simulator Boost Duchenne Muscular Dystrophy Market • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Duchenne Muscular Dystrophy Market?
Duchenne muscular dystrophy is a genetic disease that causes gradual muscle loss, affecting the skeletal, cardiac, and lung muscles. It is caused by a mutation in the gene responsible for producing dystrophin, a protein that is crucial for the healthy operation of muscles. The main types of therapies included in duchenne muscular dystrophy are mutation suppression and exon skipping approach. Exon skipping is the most prevalent type of alternative splicing, in which introns and intervals on a pre-mRNA molecule that ordinarily function as exons of a gene are removed together when a final mRNA is formed. therapeutic classes included are molecular-based therapies and steroid therapy and the route of administration are oral, parenteral, and others. These are distributed through hospital pharmacies, online pharmacies, and retail pharmacies for use in end-users such as hospitals, homecare, specialty clinics, and others.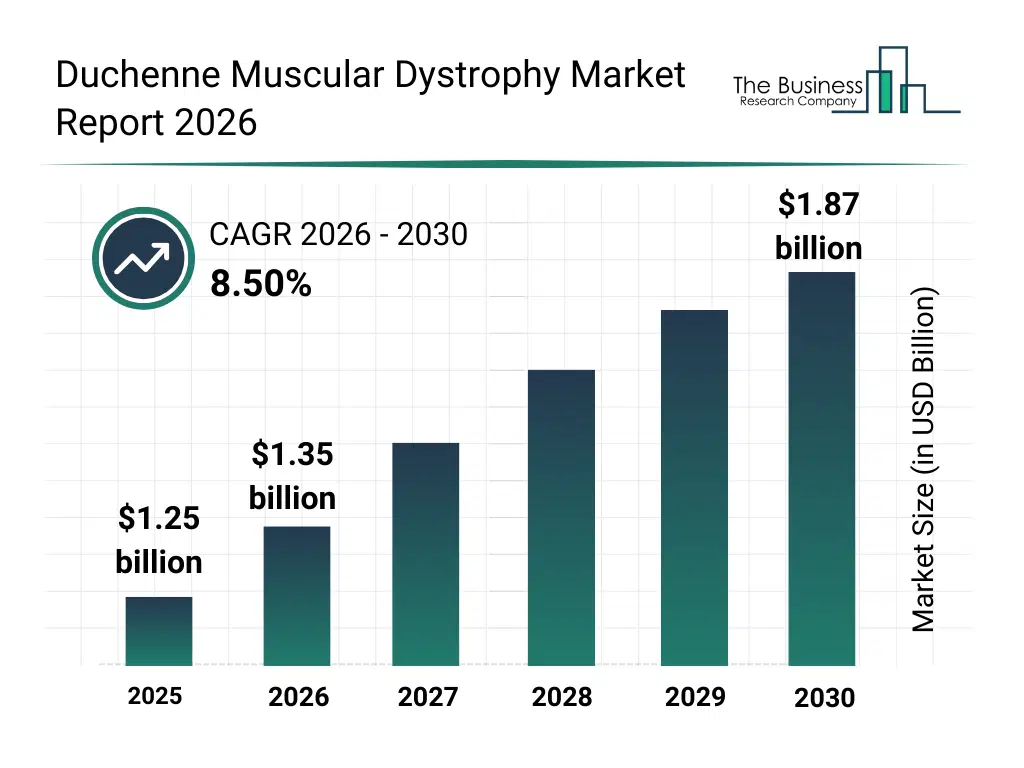
What Is The Duchenne Muscular Dystrophy Market Size and Share 2026?
The duchenne muscular dystrophy market size has grown strongly in recent years. It will grow from $1.25 billion in 2025 to $1.35 billion in 2026 at a compound annual growth rate (CAGR) of 7.9%. The growth in the historic period can be attributed to increasing prevalence of duchenne muscular dystrophy, advancements in molecular biology techniques, rising awareness about rare genetic disorders, development of early diagnostic tools, increasing research collaborations between academic and pharmaceutical institutions.What Is The Duchenne Muscular Dystrophy Market Growth Forecast?
The duchenne muscular dystrophy market size is expected to see strong growth in the next few years. It will grow to $1.87 billion in 2030 at a compound annual growth rate (CAGR) of 8.5%. The growth in the forecast period can be attributed to growth in gene therapy adoption, expansion of precision medicine approaches, increased investment in rare disease drug development, regulatory incentives for orphan drugs, integration of digital health solutions in patient management. Major trends in the forecast period include gene editing advancements, personalized treatment protocols, rare disease research funding, patient-centric care models, orphan drug development.Global Duchenne Muscular Dystrophy Market Segmentation
1) By Therapy: Mutation Suppression, Exon Skipping Approach 2) By Therapeutic Class: Molecular Based Therapies, Steroid Therapy 3) By Route Of Administration: Oral, Parenteral, Other Routes Of Administration 4) By Distribution Channel: Hospital Pharmacy, Online Pharmacy, Retail Pharmacy 5) By End User: Hospitals, Homecare, Specialty Clinics, Other End-Users Subsegments: 1) By Mutation Suppression: Antisense Oligonucleotides (ASOs), Small Molecules, Gene Therapy 2) By Exon Skipping Approach: Exon-Skipping Therapies, Combination Therapies, Clinical Trials And Investigational DrugsWhat Is The Driver Of The Duchenne Muscular Dystrophy Market?
The increasing prevalence of chronic diseases is expected to propel the growth of the Duchenne muscular dystrophy market in the future. A chronic disease is a health issue or condition with long-lasting symptoms lasting for at least a year. The Duchenne muscular dystrophy contributes to the prevalence of chronic diseases by addressing the specific therapeutic needs of individuals with this genetic disorder, highlighting the broader significance of personalized medicine in managing and potentially mitigating the impact of chronic conditions. For instance, in January 2023, according to the National Library of Medicine, a US-based government agency, the number of people in the US aged 50 and older who have at least 1 chronic condition would increase to 142.66 million by 2050. Therefore, the increasing prevalence of chronic diseases is driving the growth of the Duchenne muscular dystrophy industry.Key Players In The Global Duchenne Muscular Dystrophy Market
Major companies operating in the duchenne muscular dystrophy market are Pfizer Inc., GlaxoSmithKline plc, BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., PTC Therapeutics Inc., Asklepios BioPharmaceutical Inc., Catabasis Pharmaceuticals Inc., Wave Life Sciences Ltd., Bristol Myers Squibb Company, BridgeBio Pharma Inc., Rocket Pharmaceuticals Inc., Italfarmaco SpA, Solid Biosciences Inc., Avidity Biosciences Inc., Fulcrum Therapeutics Inc., Akashi Therapeutics Inc., Capricor Therapeutics Inc., Santhera Pharmaceuticals Holding, Armagen Technologies Inc., Dyne Therapeutics Inc., Benitec Biopharma Limited, Edgewise Therapeutics Inc., WAVE Life Sciences Ltd.Global Duchenne Muscular Dystrophy Market Trends and Insights
Major companies operating in the Duchenne muscular dystrophy market are developing technological advancements, such as clinical trial design, to enhance the efficiency and effectiveness of drug development processes, improve patient outcomes, and accelerate the approval of new therapies. Clinical trial design is the process of organizing and planning a clinical trial to evaluate a new treatment's safety and effectiveness, including defining goals, selecting participants, and outlining methods and analyses. For instance, in March 2024, Critical Path Institute (C-Path), a US-based non-profit organization, launched the DMD Clinical Trial Simulator (CTS). This innovative tool, developed by C-Path's Duchenne Regulatory Science Consortium, aims to optimize the design of efficacy studies for DMD therapies. Key features of the CTS include advanced modeling of functional test performance, the capability to simulate various scenarios, increased efficiency in trial design, and public accessibility via the C-Path website and the Rare Disease Cures Accelerator-Data and Analytics Platform (RDCA-DAP). As such, it serves as a valuable resource for researchers and pharmaceutical companies engaged in DMD research.What Are Latest Mergers And Acquisitions In The Duchenne Muscular Dystrophy Market?
In October 2025, Novartis AG., a Switzerland-based global pharmaceutical company, acquired Avidity Biosciences, Inc., for $12 billion in cash. With this acquisition, Novartis aims to expand its RNA-targeted therapies portfolio, accelerate its presence in genetic disease treatments, and enhance its ability to develop innovative medicines for patients with serious and rare conditions. Avidity Biosciences, Inc., is a US-based biotechnology company that developing a treatment for Duchenne Muscular Dystrophy (DMD) called del-zota.Regional Insights
North America was the largest region in the Duchenne muscular dystrophy market in 2025. Asia-Pacific is expected to be the fastest-growing region in the global duchenne muscular dystrophy market during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Duchenne Muscular Dystrophy Market?
The duchenne muscular dystrophy market includes revenues earned by entities by providing respiratory therapy, physical therapy and gene therapy. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Duchenne Muscular Dystrophy Market Report 2026?
The duchenne muscular dystrophy market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the duchenne muscular dystrophy industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Duchenne Muscular Dystrophy Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.35 billion |
| Revenue Forecast In 2035 | $1.87 billion |
| Growth Rate | CAGR of 7.9% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Therapy, Therapeutic Class, Route Of Administration, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., GlaxoSmithKline plc, BioMarin Pharmaceutical Inc., Sarepta Therapeutics Inc., PTC Therapeutics Inc., Asklepios BioPharmaceutical Inc., Catabasis Pharmaceuticals Inc., Wave Life Sciences Ltd., Bristol Myers Squibb Company, BridgeBio Pharma Inc., Rocket Pharmaceuticals Inc., Italfarmaco SpA, Solid Biosciences Inc., Avidity Biosciences Inc., Fulcrum Therapeutics Inc., Akashi Therapeutics Inc., Capricor Therapeutics Inc., Santhera Pharmaceuticals Holding, Armagen Technologies Inc., Dyne Therapeutics Inc., Benitec Biopharma Limited, Edgewise Therapeutics Inc., WAVE Life Sciences Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
