
Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Report 2026
Global Outlook – By Therapies (Nutritional Management, Pancreatic Enzyme Replacement Therapy (PERT), Lifestyle Modifications Approach), By Diagnostics (Blood Tests, Magnetic Resonance Imaging (MRI), Endoscopic Ultra-Sonography (EUS), Computerized Tomography (CT) Scanning), By Drug Type (Generic, Branded), By Distribution Channel (Direct Tender, Retail Pharmacy , Third-Party Distributor, Other Distribution Channels), By End User (Hospitals, Specialty Clinics, Homecare, Diagnostic Center, Research And Academic Institutes, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Overview
• Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics market size has reached to $6.11 billion in 2025 • Expected to grow to $8.97 billion in 2030 at a compound annual growth rate (CAGR) of 8.2% • Growth Driver: Rising Diabetes Prevalence Fuels Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market • Market Trend: Strategic Partnerships Drive Innovation In Exocrine Pancreatic Insufficiency (EPI) • North America was the largest region in 2025.What Is Covered Under Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market?
Exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics refer to treating a condition in which the pancreas does not produce enough digestive enzymes required for the proper breakdown and absorption of nutrients from food in the small intestine. EPI diagnostics and therapeutics play a crucial role in diagnosing and managing exocrine pancreatic insufficiency, helping individuals with EPI achieve better nutrient absorption and overall well-being. The main types of therapies included in exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics are nutritional management, pancreatic enzyme replacement therapy (PERT) and lifestyle modifications approach. Nutritional management is a specialized approach to healthcare that involves the use of nutrition and diet to treat and manage various medical and health conditions. Various types of diagnostics are involved, including blood tests, magnetic resonance imaging (MRI), endoscopic ultrasonography (EUS) and computerized tomography (CT) scanning. Several types of drugs are used, such as generic and branded, which are sold through various distribution channels, including direct tender, retail pharmacy, third-party distributor and others and are used by various end users, such as hospitals, specialty clinics, homecare, diagnostic centers, research and academic institutes and others.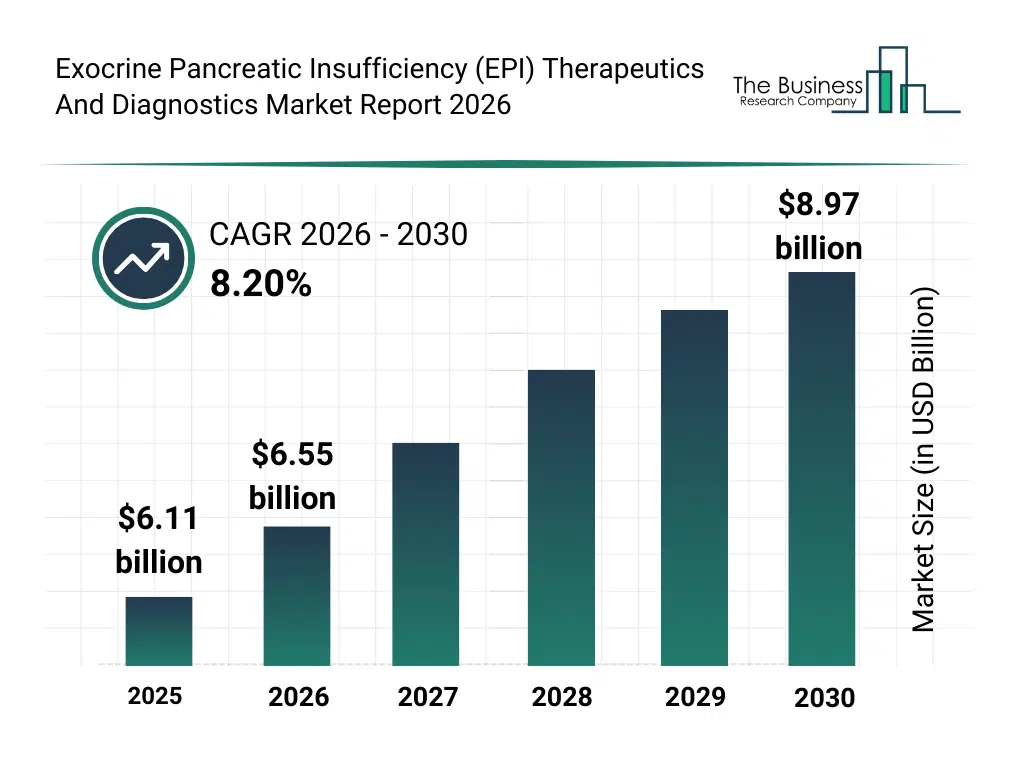
What Is The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Size and Share 2026?
The exocrine pancreatic insufficiency (epi) therapeutics and diagnostics market size has grown strongly in recent years. It will grow from $6.11 billion in 2025 to $6.55 billion in 2026 at a compound annual growth rate (CAGR) of 7.1%. The growth in the historic period can be attributed to increasing prevalence of chronic pancreatitis, rising awareness about digestive disorders, limited access to specialized diagnostics, adoption of nutritional supplements, growth in home healthcare services.What Is The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Growth Forecast?
The exocrine pancreatic insufficiency (epi) therapeutics and diagnostics market size is expected to see strong growth in the next few years. It will grow to $8.97 billion in 2030 at a compound annual growth rate (CAGR) of 8.2%. The growth in the forecast period can be attributed to advancements in personalized enzyme therapies, integration of ai in diagnostics, rising demand for non-invasive imaging, expansion of telehealth services, growing focus on patient-centric nutritional management. Major trends in the forecast period include advanced pancreatic enzyme replacement therapies, point-of-care diagnostic solutions, personalized nutritional management programs, non-invasive imaging techniques for epi, integration of telehealth in epi management.Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Segmentation
1) By Therapies: Nutritional Management, Pancreatic Enzyme Replacement Therapy (PERT), Lifestyle Modifications Approach 2) By Diagnostics: Blood Tests, Magnetic Resonance Imaging (MRI), Endoscopic Ultra-Sonography (EUS), Computerized Tomography (CT) Scanning 3) By Drug Type: Generic, Branded 4) By Distribution Channel: Direct Tender, Retail Pharmacy , Third-Party Distributor, Other Distribution Channels 5) By End User: Hospitals, Specialty Clinics, Homecare, Diagnostic Center, Research And Academic Institutes, Other End Users Subsegments: 1) By Nutritional Management: Dietary Modifications, Nutritional Supplements 2) By Pancreatic Enzyme Replacement Therapy (PERT): Capsule Formulations, Tablet Formulations, Powder Formulations 3) By Lifestyle Modifications Approach: Weight Management, Physical Activity Recommendations, Smoking Cessation ProgramsWhat Are The Drivers Of The Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market?
The increasing number of people with diabetes is expected to propel the growth of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market going forward. Diabetes refers to a chronic metabolic disorder characterized by high blood sugar levels (hyperglycemia) over a prolonged period. This condition results from the body's inability to produce enough insulin or effectively use the insulin it produces. EPI therapy, which involves pancreatic enzyme replacement therapy (PERT), can help improve nutrient absorption in people with diabetes and EPI. It can also help improve glycemic control in people with diabetes. For instance, in June 2024, according to The National Health Service (NHS England), a UK-based publicly funded healthcare system, in 2023, the number of individuals identified by the NHS in England as having pre-diabetes rose to 3,615,330, marking an 18% increase from 3,065,825 in 2022. Additionally, among those under 40, cases surged nearly 25%, growing from 173,166 in 2022 to 216,440 in 2023. Therefore, the increasing number of people with diabetes is driving the growth of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics industry. The increasing incidence of gastrointestinal diseases is expected to propel the growth of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market going forward. Gastrointestinal diseases refer to a group of medical conditions that affect the gastrointestinal tract, which includes the organs responsible for digestion. Exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics play a crucial role in managing gastrointestinal diseases by addressing the underlying enzyme deficiency and aiding in the proper digestion of nutrients. Exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics play a crucial role in the management of gastrointestinal diseases, as they address the underlying enzyme deficiency and assist in digestion. For instance, in June 2023, according to Crohn's and Colitis Canada, a Canada-based non-profit organization, over 322,600 Canadians were estimated to be living with inflammatory bowel diseases (IBD) in 2023, accounting for approximately 0.82% of the population. As the prevalence of IBD continues, it is estimated that around 470,000 Canadians will be living with IBD by 2035, approximately 1.1% of the population, or 1 in every 91 people in the country. Therefore, the increasing incidence of gastrointestinal diseases is expected to propel the growth of the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics industry.Key Players In The Global Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market
Major companies operating in the exocrine pancreatic insufficiency (epi) therapeutics and diagnostics market are Abbott Laboratories, Metagenics LLC, Chiesi Farmaceutici S.p.A., Nordmark Arzneimittel GmbH & Co. KG, Digestive Care Inc., Bioserv Diagnostics Gmbh, Aptalis Pharma Inc., EagleBiosciences Inc., ScheBo Biotech AG, Solvay S.A., Codexis Inc., ChiRhoClin Inc., Alcresta Therapeutics Inc., Cilian AG, First Wave BioPharma Inc., Laboratory Corporation of America Holdings, Organon, Vivus Inc., Anthera Pharmaceuticals Inc., Teva Pharmaceutical Industries LtdGlobal Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Trends and Insights
Major companies operating in the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market are adopting a strategic partnership approach to enhance technology integration and expand market reach. A strategic partnership typically refers to a collaborative relationship between two or more organizations where they combine their resources, expertise, and efforts to achieve common goals or objectives. For instance, in February 2023, Codexis, Inc., a US-based protein engineering company, and Nestlé Health Science, a Switzerland-based science of nutrition, announced interim results from their Phase 1 clinical trial of CDX-7108, an investigational therapy aimed at treating exocrine pancreatic insufficiency (EPI). The results from this Phase 1 trial pave the way for further studies that could significantly improve treatment options for patients suffering from EPI.Regional Insights
North America was the largest region in the exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market?
The exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market consists of revenues earned by entities by providing dietary interventions and bile acid replacement therapy (BAR). The market value includes the value of related goods sold by the service provider or included within the service offering. The exocrine pancreatic insufficiency (EPI) therapeutics and diagnostics market also includes sales of Creon, Zenpep and Ultresa. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Report 2026?
The exocrine pancreatic insufficiency (epi) therapeutics and diagnostics market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the exocrine pancreatic insufficiency (epi) therapeutics and diagnostics industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Exocrine Pancreatic Insufficiency (EPI) Therapeutics And Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.55 billion |
| Revenue Forecast In 2035 | $8.97 billion |
| Growth Rate | CAGR of 7.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Therapies, Diagnostics, Drug Type, Distribution Channel, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, Metagenics LLC, Chiesi Farmaceutici S.p.A., Nordmark Arzneimittel GmbH & Co. KG, Digestive Care Inc., Bioserv Diagnostics Gmbh, Aptalis Pharma Inc., EagleBiosciences Inc., ScheBo Biotech AG, Solvay S.A., Codexis Inc., ChiRhoClin Inc., Alcresta Therapeutics Inc., Cilian AG, First Wave BioPharma Inc., Laboratory Corporation of America Holdings, Organon, Vivus Inc., Anthera Pharmaceuticals Inc., Teva Pharmaceutical Industries Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
