
Hemostasis Diagnostic Devices And Equipment Market Report 2026
Global Outlook – By Product (Analyzers, Coagulation Instrument, Other Hemostasis Instruments, Reagents and Kits), By Device Technology (Automated, Semi-Automated, Manual), By End User (Hospitals, Clinics, Independent Diagnostic, Laboratories, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Hemostasis Diagnostic Devices And Equipment Market Overview
• Hemostasis Diagnostic Devices And Equipment market size has reached to $5.66 billion in 2025 • Expected to grow to $10.12 billion in 2030 at a compound annual growth rate (CAGR) of 12% • Growth Driver: Rising Number Of Surgical Procedures Driving Market Growth Due To Increased Demand For Hemostasis Monitoring Solutions • Market Trend: Embedding Factor Xa Inhibitor Coagulation Testing For Personalized Anticoagulation Management • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Hemostasis Diagnostic Devices And Equipment Market?
Hemostasis diagnostic devices and equipment are specialized tools used to assess the blood clotting process and diagnose bleeding disorders. These devices help healthcare professionals monitor and evaluate various aspects of hemostasis, such as clot formation and dissolution. The main products of hemostasis diagnostic devices and equipment are analyzers, coagulation instruments, other hemostasis instruments, and reagents and kits. A hemostasis diagnostic analyzer is a tool utilized to predict the cause of bleeding during surgery by identifying any hemostatic defects. The hemostasis diagnostic devices and equipment are automated, semi-automated, and manual and are used by hospitals, clinics, independent diagnostic, laboratories, and others.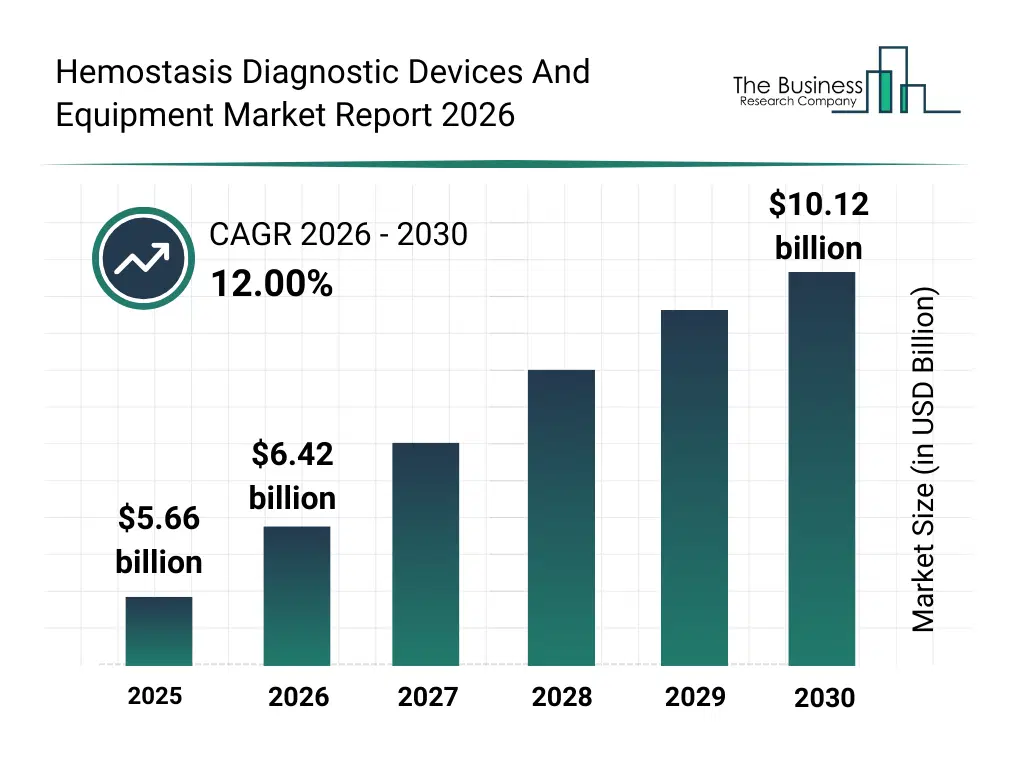
What Is The Hemostasis Diagnostic Devices And Equipment Market Size and Share 2026?
The hemostasis diagnostic devices and equipment market size has grown rapidly in recent years. It will grow from $5.66 billion in 2025 to $6.42 billion in 2026 at a compound annual growth rate (CAGR) of 13.4%. The growth in the historic period can be attributed to increasing incidence of bleeding disorders, expansion of surgical procedures requiring coagulation monitoring, growth of hospital laboratory services, adoption of standardized clotting tests, availability of advanced coagulation reagents.What Is The Hemostasis Diagnostic Devices And Equipment Market Growth Forecast?
The hemostasis diagnostic devices and equipment market size is expected to see rapid growth in the next few years. It will grow to $10.12 billion in 2030 at a compound annual growth rate (CAGR) of 12.0%. The growth in the forecast period can be attributed to increasing demand for rapid bedside diagnostics, rising adoption of AI-assisted coagulation analysis, expansion of personalized anticoagulant therapy, growing investments in advanced laboratory infrastructure, increasing focus on preventive diagnostics. Major trends in the forecast period include increasing deployment of automated coagulation analyzers, rising demand for point-of-care hemostasis testing, growing integration of digital result interpretation tools, expansion of comprehensive clotting analysis systems, enhanced focus on rapid diagnostic turnaround.Global Hemostasis Diagnostic Devices And Equipment Market Segmentation
1) By Product: Analyzers, Coagulation Instrument, Other Hemostasis Instruments, Reagents and Kits 2) By Device Technology: Automated, Semi-Automated, Manual 3) By End User: Hospitals, Clinics, Independent Diagnostic, Laboratories, Other End Users Subsegments: 1) By Analyzers: Hematology Analyzers, Platelet Function Analyzers, Thromboelastography (TEG) Analyzers 2) By Coagulation Instruments: Prothrombin Time (PT) Instruments, Activated Partial Thromboplastin Time (aPTT) Instruments, Fibrinogen Testing Instruments, Thrombin Time (TT) Instruments 3) By Other Hemostasis Instruments: Blood Clotting Time (BCT) Instruments, Bleeding Time (BT) Test Devices 4) By Reagents and Kits: Coagulation Reagents, Blood Collection Kits, Point-of-care Testing Kits, Thrombosis KitsWhat Is The Driver Of The Hemostasis Diagnostic Devices And Equipment Market?
The rising number of surgical procedures is expected to propel the growth of hemostasis diagnostic devices and equipment market going forward. Surgical procedures refer to medical interventions or treatments that involve making incisions or using invasive techniques to access and treat internal or external areas of the body. The rise in surgical procedures is due to the aging population, which increases the prevalence of chronic and complex medical conditions requiring surgical intervention. Surgical procedures benefit significantly from the use of hemostasis diagnostic devices by improving the management of blood clotting, minimizing risks, and enhancing overall surgical efficiency, ultimately leading to better patient outcomes. For instance, in April 2025, according to the British Broadcasting Corporation, a UK-based public service broadcaster, approximately 27,462 cosmetic surgical procedures were performed in 2024, representing a 5 percent increase from 2023, with more than nine in ten procedures performed on women. Therefore, the rising number of surgical procedures drives the growth of the hemostasis diagnostic devices and equipment industry going forward.Key Players In The Global Hemostasis Diagnostic Devices And Equipment Market
Major companies operating in the hemostasis diagnostic devices and equipment market are Baxter International Inc., Johnson & Johnson, Abbott Laboratories, Beckman Coulter Inc., Becton Dickinson and Company, Chrono-Log Corporation, F. Hoffmann-La Roche Ltd., Grifols S.A., Siemens AG, Thermo Fisher Scientific Inc., Trinity Biotech plc., Ethicon Inc., B. Braun Interventional Systems Inc., Bio-Rad Laboratories Inc., CareFusion Corporation, Danaher Corporation, Diagnostica Stago SAS, Haemonetics Corporation, QuidelOrtho Corp, STAGOGlobal Hemostasis Diagnostic Devices And Equipment Market Trends and Insights
Major companies operating in the hemostasis diagnostic devices and equipment market are focusing on technological development, such as inhibitor-specific coagulation testing, to improve patient safety, optimize anticoagulation therapy, and reduce the risk of bleeding or thrombotic events in individuals on direct oral anticoagulants (DOACs). These tests enable laboratories and clinicians to accurately measure anticoagulant activity, guide dosage adjustments, and support personalized therapy management. For instance, in February 2024, Roche Diagnostics, a Switzerland-based manufacturer of diagnostic equipment and reagents for research and medical diagnostic applications, launched three new Factor Xa inhibitor coagulation tests for their cobas platforms, designed to measure apixaban, rivaroxaban, and edoxaban activity in patient plasma samples. The tests are intended to provide rapid, accurate, and reliable results to guide clinical decision-making in situations such as surgery, emergency care, or suspected bleeding complications. This launch reflects the market trend toward personalized anticoagulation management, enhancing safety and efficacy for patients receiving direct Factor Xa inhibitor therapy while supporting laboratories with standardized, automated assay solutions.What Are Latest Mergers And Acquisitions In The Hemostasis Diagnostic Devices And Equipment Market?
In March 2023, Werfen, S.A., a Europe-based company that manufactures diagnostic instruments and reagents for hospitals and laboratories, acquired Immucor, Inc. for an undisclosed amount. With this acquisition, Werfen, S.A. aims to enhance its diagnostic solutions and improve patient care. Immucor Inc. is a US-based company that manufactures diagnostic products and systems primarily for blood transfusion and immunohematology.Regional Outlook
North America was the largest region in the global hemostasis diagnostic devices and equipment market in 2025. Asia-Pacific was the second-largest region in the hemostasis diagnostic devices and equipment market. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are China, India, Japan, Australia, Indonesia, South Korea, Bangladesh, Thailand, Vietnam, Malaysia, Singapore, Philippines, Hong Kong, Taiwan, New Zealand, UK, Germany, France, Italy, Spain, Austria, Belgium, Denmark, Finland, Ireland, Netherlands, Norway, Portugal, Sweden, Switzerland, Russia, Czech Republic, Poland, Romania, Ukraine, USA, Canada, Mexico, Brazil, Chile, Argentina, Colombia, Peru, Saudi Arabia, Israel, Iran, Turkey, UAE, Egypt, Nigeria, South Africa.What Defines the Hemostasis Diagnostic Devices And Equipment Market?
Hemostasis diagnostic devices and equipment consist of sales of instruments such as analyzers, coagulation instruments, other hemostasis instruments, and reagents and kits that are used in hemostasis diagnostics. Values in this market are factory gate values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Hemostasis Diagnostic Devices And Equipment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.42 billion |
| Revenue Forecast In 2035 | $10.12 billion |
| Growth Rate | CAGR of 13.4% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Device Technology, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Baxter International Inc., Johnson & Johnson, Abbott Laboratories, Beckman Coulter Inc., Becton Dickinson and Company, Chrono-Log Corporation, F. Hoffmann-La Roche Ltd., Grifols S.A., Siemens AG, Thermo Fisher Scientific Inc., Trinity Biotech plc., Ethicon Inc., B. Braun Interventional Systems Inc., Bio-Rad Laboratories Inc., CareFusion Corporation, Danaher Corporation, Diagnostica Stago SAS, Haemonetics Corporation, QuidelOrtho Corp, STAGO |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
