
Liver Cancer Diagnostics Market Report 2026
Global Outlook – By Type (Hepatocellular Carcinoma, Cholangiocarcinoma, Hepatoblastoma), By Screening (Laboratory Testing, Imaging, Biopsy, Other Screenings), By Technology (Fluorescent In Situ Hybridization (FISH), Immunohistochemical (IHC), Other Technologies), By End Use (Hospitals And Diagnostic Laboratories, Academic And Research Institutes) – Market Size, Trends, Strategies, and Forecast to 2035
Liver Cancer Diagnostics Market Overview
• Liver Cancer Diagnostics market size has reached to $10.32 billion in 2025 • Expected to grow to $14.75 billion in 2030 at a compound annual growth rate (CAGR) of 7.4% • Growth Driver: Surge In Liver Cancer Cases Fueling The Growth Of The Market Due To Rising Prevalence And Mortality Rates • Market Trend: Advanced Biomarker-Based Assays Launched To Improve Liver Cancer Diagnostics • North America was the largest region in 2025.What Is Covered Under Liver Cancer Diagnostics Market?
Liver cancer diagnostics refers to the various methods employed to detect and diagnose liver cancer, a type of cancer that originates in the liver. It is used for the early detection and diagnosis of liver cancer via imaging and molecular testing. The main types of cancer included in liver cancer diagnostics are hepatocellular carcinoma, cholangiocarcinoma, hepatoblastoma, and others. Hepatocellular carcinoma (HCC) refers to the primary liver cancer that most commonly affects people who have cirrhosis and preexisting chronic liver disease. The several types of screening are involved are laboratory testing, imaging, endoscopy, biopsy, and others. The various types of technology are included, such as fluorescent in situ hybridization (FISH), comparative genomic hybridization (CGH), immunohistochemical (IHC), and others, which are used in several end uses, such as hospitals and diagnostic laboratories, academic and research institutes, and pharmaceutical and CRO laboratories.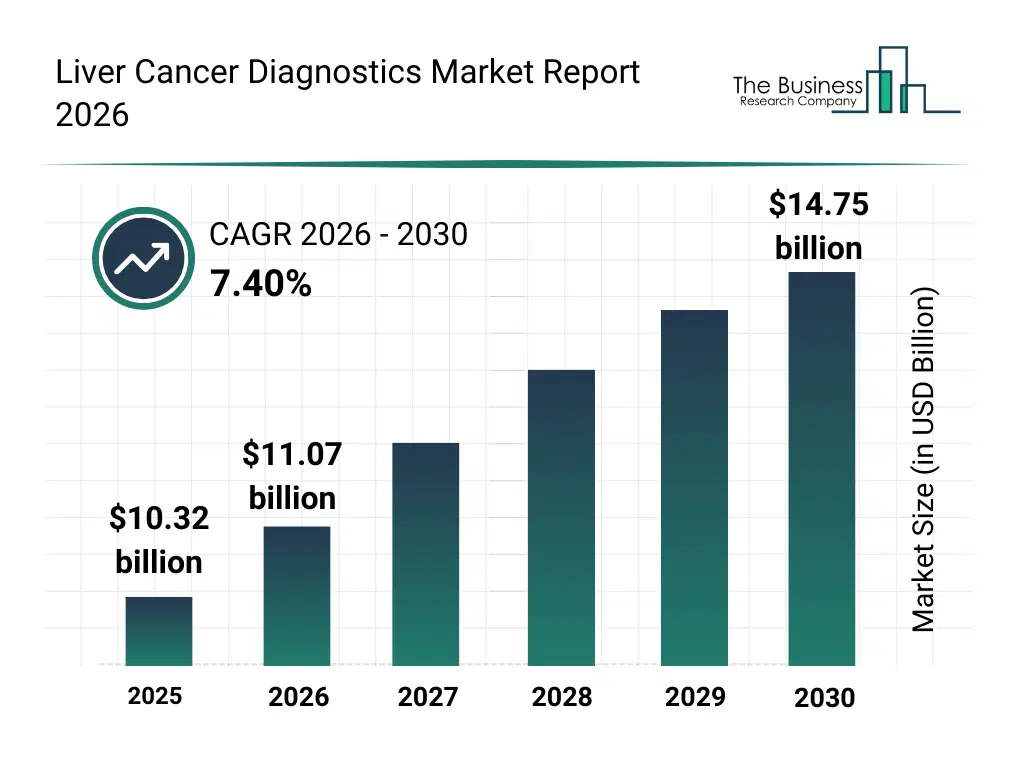
What Is The Liver Cancer Diagnostics Market Size and Share 2026?
The liver cancer diagnostics market size has grown strongly in recent years. It will grow from $10.32 billion in 2025 to $11.07 billion in 2026 at a compound annual growth rate (CAGR) of 7.3%. The growth in the historic period can be attributed to increasing prevalence of chronic liver diseases, rising incidence of hepatocellular carcinoma, expansion of hospital diagnostic infrastructure, growing awareness of early cancer screening, advancements in imaging and pathology techniques.What Is The Liver Cancer Diagnostics Market Growth Forecast?
The liver cancer diagnostics market size is expected to see strong growth in the next few years. It will grow to $14.75 billion in 2030 at a compound annual growth rate (CAGR) of 7.4%. The growth in the forecast period can be attributed to increasing adoption of precision oncology diagnostics, rising demand for non-invasive diagnostic methods, expansion of molecular testing capabilities, growing investments in oncology research, increasing use of ai-assisted diagnostic platforms. Major trends in the forecast period include increasing adoption of advanced molecular diagnostic tests, rising use of imaging-based early detection tools, growing integration of biomarker-based screening methods, expansion of personalized oncology diagnostics, enhanced focus on early-stage cancer detection.Global Liver Cancer Diagnostics Market Segmentation
1) By Type: Hepatocellular Carcinoma, Cholangiocarcinoma, Hepatoblastoma 2) By Screening: Laboratory Testing, Imaging, Biopsy, Other Screenings 3) By Technology: Fluorescent In Situ Hybridization (FISH), Immunohistochemical (IHC), Other Technologies 4) By End Use: Hospitals And Diagnostic Laboratories, Academic And Research Institutes Subsegments: 1) By Hepatocellular Carcinoma: Early-Stage Hepatocellular Carcinoma, Advanced-Stage Hepatocellular Carcinoma 2) By Cholangiocarcinoma: Intrahepatic Cholangiocarcinoma, Extrahepatic Cholangiocarcinoma 3) By Hepatoblastoma: Pure Hepatoblastoma, Mixed HepatoblastomaWhat Is The Driver Of The Liver Cancer Diagnostics Market?
The rising prevalence of liver cancer is expected to propel the growth of the liver cancer diagnostics market going forward. Liver cancer refers to a condition in which the liver cells grow and multiply out of control, giving rise to a life-threatening illness. Liver cancer diagnostic tests are performed to determine whether cancer cells are spreading within the liver or surrounding it after the diagnosis of primary carcinoma of the liver, so the rising prevalence of liver cancer boosts the liver cancer diagnostics market. For instance, in January 2023, according to the Cancer Facts & Figures 2023 report published by the American Cancer Society, a US-based voluntary health organization, the number of new cases of liver and intrahepatic bile duct cancer in US females reached 13,230 in 2023. Furthermore, in 2023, an estimated 41,210 new cases of liver cancer were diagnosed in the US, where 29,380 people are expected to die. Therefore, the rising prevalence of liver cancer is driving the growth of the liver cancer diagnostic industry.Key Players In The Global Liver Cancer Diagnostics Market
Major companies operating in the liver cancer diagnostics market are Abbott Laboratories, F Hoffmann La Roche Ltd, Qiagen NV, Thermo Fisher Scientific Inc, Siemens Healthineers, Illumina Inc, Koninklijke Philips NV, Epigenomics AG, Digna Biotech SL, Biocept Inc, Danaher Corporation, Foundation Medicine Inc, Agilent Technologies Inc, Genemed Biotechnologies Inc, Merck KGaA, Fujifilm Medical Systems USA Inc, GE Healthcare, Advanced Cell Diagnostics Inc, Oxford Gene Technology, Leica Biosystems Nussloch GmbH, Exiqon AS, BioGenex Laboratories Inc, OncoCyte Corporation, Guardant Health Inc, Exact Sciences Corporation, Grail Inc, Freenome Holdings Inc, Natera Inc, Personal Genome Diagnostics Inc, Thrive Earlier Detection CorpGlobal Liver Cancer Diagnostics Market Trends and Insights
Major companies operating in the liver cancer diagnostics market are developing advanced biomarker-based assays and algorithms to improve diagnostic accuracy and support early detection. Biomarker-based assays and algorithms refer to diagnostic tools that utilize specific biological markers to identify diseases, providing more precise and efficient diagnostic capabilities. For instance, in November 2023, Roche Diagnostics, a US-based biotech corporation, launched the Elecsys PIVKA-II assay and Elecsys GAAD algorithm. The Elecsys PIVKA-II assay detects elevated levels of the PIVKA-II biomarker, which is associated with hepatocellular carcinoma (HCC), improving early detection of liver cancer in high-risk patients. The Elecsys GAAD algorithm integrates multiple diagnostic markers, such as PIVKA-II and AFP, to enhance diagnostic precision and streamline decision-making in liver cancer detection.What Are Latest Mergers And Acquisitions In The Liver Cancer Diagnostics Market?
In December 2024, Metropolis Healthcare Ltd., an India-based healthcare diagnostics company, acquired Core Diagnostics for $0.0278 billion. With this acquisition, Metropolis aims to significantly enhance its advanced cancer-testing capabilities and expand its presence in north and east India, strengthening its leadership in the specialized diagnostics space. CORE Diagnostics Private Limited is a India-based specialty diagnostics company that provide provides tests for liver cancer diagnostics.Regional Outlook
North America was the largest region in the liver cancer diagnostics market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Liver Cancer Diagnostics Market?
The liver cancer diagnostics market includes revenues earned by entities by liver transplants, CT scans, and liver function tests. The market value includes the value of related goods sold by the service provider or included within the service offering. The liver cancer diagnostics market also includes sales of In Vitro and Imaging Tools, AI tools, and ML tools which are used in providing liver cancer diagnostic services. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Liver Cancer Diagnostics Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $11.07 billion |
| Revenue Forecast In 2035 | $14.75 billion |
| Growth Rate | CAGR of 7.3% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Screening, Technology, End Use |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Abbott Laboratories, F Hoffmann La Roche Ltd, Qiagen NV, Thermo Fisher Scientific Inc, Siemens Healthineers, Illumina Inc, Koninklijke Philips NV, Epigenomics AG, Digna Biotech SL, Biocept Inc, Danaher Corporation, Foundation Medicine Inc, Agilent Technologies Inc, Genemed Biotechnologies Inc, Merck KGaA, Fujifilm Medical Systems USA Inc, GE Healthcare, Advanced Cell Diagnostics Inc, Oxford Gene Technology, Leica Biosystems Nussloch GmbH, Exiqon AS, BioGenex Laboratories Inc, OncoCyte Corporation, Guardant Health Inc, Exact Sciences Corporation, Grail Inc, Freenome Holdings Inc, Natera Inc, Personal Genome Diagnostics Inc, Thrive Earlier Detection Corp |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
