
Optic Atrophy Management Market Report 2026
Global Outlook – By Drug Type (Disulfiram, Amebicides, Ethambutol, Isoniazid, Chloramphenicol, Vincristine, Ciclosporin, Cimetidine), By Route Of Administration (Oral, Injectable), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By End-User (Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Rehabilitation Centers) - Market Size, Trends, And Global Forecast 2026-2035
Optic Atrophy Management Market Overview
• Optic Atrophy Management market size has reached to $8.91 billion in 2025 • Expected to grow to $12.32 billion in 2030 at a compound annual growth rate (CAGR) of 6.7% • Growth Driver: Rising Diabetes Cases Propel Demand For Optic Atrophy Solutions • Market Trend: Pioneer Precision Medicine And A Gene-Based Optic Atrophy Treatment Trial • North America was the largest region in 2025.What Is Covered Under Optic Atrophy Management Market?
Optic atrophy refers to a medical condition characterized by the damage or degeneration of the optic nerve, which is responsible for transmitting visual information from the eye to the brain. It is distinguished by the degeneration of retinal ganglion cell axons, which results in optic nerve atrophy. The treatment options may include controlling the underlying ailment, vision rehabilitation, or supportive measures to optimize residual eyesight. The main types of drugs used in optic atrophy management are disulfiram, amebicides, ethambutol, isoniazid, chloramphenicol, vincristine, ciclosporin, and cimetidine. Disulfiram refers to an alcohol-sensitizing medication that interferes with the body's ability to metabolize alcohol and is used as part of a comprehensive approach to address alcohol use disorder and its associated health risks, including optic atrophy. The treatment options are administered through oral and injectable routes, which are distributed through channels such as hospital pharmacies, retail pharmacies, and online pharmacies.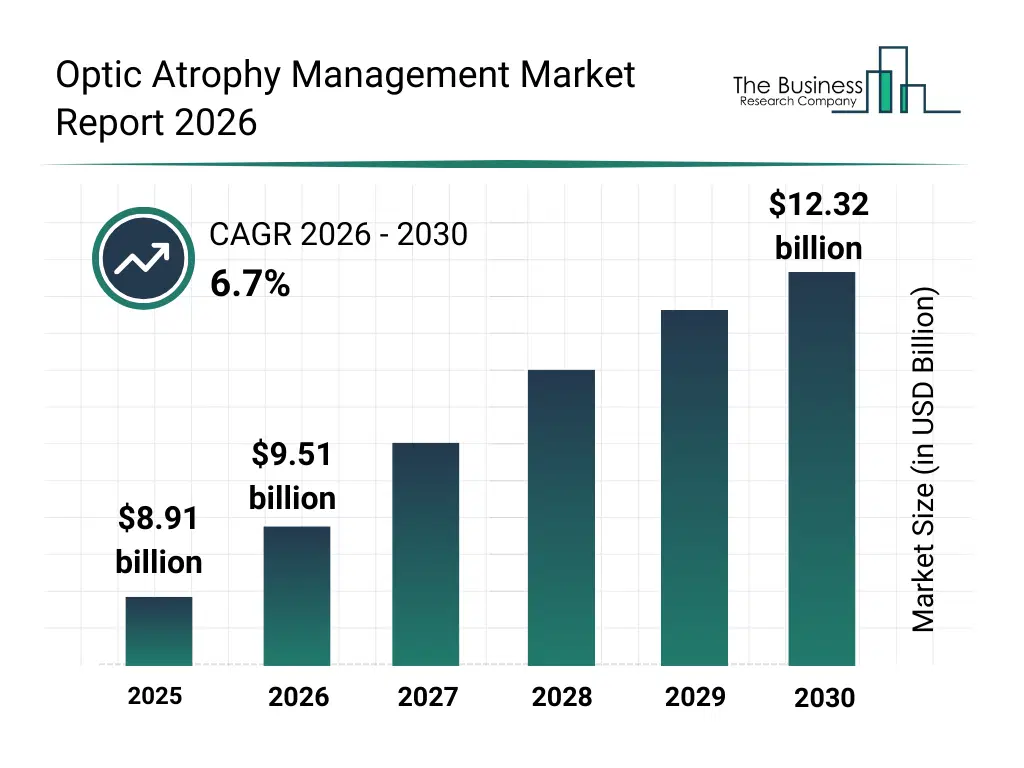
What Is The Optic Atrophy Management Market Size and Share 2026?
The optic atrophy management market size has grown strongly in recent years. It will grow from $8.91 billion in 2025 to $9.51 billion in 2026 at a compound annual growth rate (CAGR) of 6.8%. The growth in the historic period can be attributed to high prevalence of optic nerve damage due to glaucoma and ischemic disorders, limited availability of curative therapies, growing incidence of infectious and drug-induced optic neuropathy, delayed diagnosis in developing healthcare systems, reliance on symptomatic and supportive treatment approaches.What Is The Optic Atrophy Management Market Growth Forecast?
The optic atrophy management market size is expected to see strong growth in the next few years. It will grow to $12.32 billion in 2030 at a compound annual growth rate (CAGR) of 6.7%. The growth in the forecast period can be attributed to rising investment in neuro-ophthalmology research, increasing awareness of optic atrophy among clinicians, growing aging population with higher neurodegenerative risk, expansion of advanced diagnostic and imaging tools, increasing focus on personalized and long-term vision management. Major trends in the forecast period include rising focus on early diagnosis and monitoring of optic nerve degeneration, increasing adoption of vision rehabilitation and supportive care programs, growing emphasis on managing underlying neurological and systemic conditions, rising demand for neuroprotective and disease-stabilizing therapies, increasing use of multidisciplinary treatment approaches in vision care.Global Optic Atrophy Management Market Segmentation
1) By Drug Type: Disulfiram, Amebicides, Ethambutol, Isoniazid, Chloramphenicol, Vincristine, Ciclosporin, Cimetidine 2) By Route Of Administration: Oral, Injectable 3) By Distribution Channel: Hospital Pharmacies, Retail Pharmacies, Online Pharmacies 4) By End-User: Hospitals, Specialty Clinics, Ambulatory Surgical Centers, Rehabilitation Centers Subsegments: 1) By Disulfiram: Oral Disulfiram Tablets, Low-Dose Disulfiram Therapy, Adjunct Neuroprotective Disulfiram Use 2) By Amebicides: Metronidazole-Based Amebicides, Tinidazole-Based Amebicides, Combination Amebicidal Therapy 3) By Ethambutol: Low-Dose Ethambutol Regimens, Dose-Adjusted Ethambutol Therapy, Ethambutol With Visual Monitoring Protocols 4) By Isoniazid: Standard Isoniazid Therapy, Pyridoxine-Supplemented Isoniazid Therapy, Modified-Dose Isoniazid Regimens 5) By Chloramphenicol: Oral Chloramphenicol Therapy, Injectable Chloramphenicol Therapy, Short-Term Chloramphenicol Use 6) By Vincristine: Low-Dose Vincristine Therapy, Intravenous Vincristine Administration, Combination Chemotherapy Regimens With Vincristine 7) By Ciclosporin: Oral Ciclosporin Therapy, Immunomodulatory Ciclosporin Regimens, Dose-Titrated Ciclosporin Treatment 8) By Cimetidine: Oral Cimetidine Therapy, Adjunctive Cimetidine Use, Dose-Adjusted Cimetidine RegimensWhat Is The Driver Of The Optic Atrophy Management Market?
An increase in the incidence of diabetes is expected to propel the growth of the optic atrophy management market going forward. Diabetes is a chronic condition characterized by elevated blood glucose levels due to insufficient insulin production or ineffective use of insulin in the body. Optic atrophy management is beneficial for diabetics, particularly those with conditions such as Wolfram syndrome, as it addresses progressive vision loss associated with nerve degeneration to prevent or slow the progression of diabetic eye complications. For instance, in February 2023, according to Diabetes Canada, a Canada-based non-profit organization, in Canada, the estimated prevalence of diabetes is projected to increase from 2,409,000 cases (15% of the population) in 2023 to 3,009,000 cases (17% of the population) by 2033. Therefore, an increase in the incidence of diabetes is driving the growth of the optic atrophy management industry.Key Players In The Global Optic Atrophy Management Market
Major companies operating in the optic atrophy management market are Pfizer Inc., Johnson And Johnson, F. Hoffmann-La Roche AG, AbbVie Inc., Bayer AG, Novartis AG, Bristol-Myers Squibb Company, Sanofi SA, GlaxoSmithKline plc, Eli Lilly And Company, Boehringer Ingelheim International GmbH., Teva Pharmaceuticals, Viatris (Mylan N.V.), Astellas Pharma Inc, Bausch Health Companies Inc., Sumitomo Dainippon Pharma Co. Ltd, Bausch And Lomb Inc., Aurobindo Pharma, Cipla Ltd, Apotex Inc. , Zydus Cadila, Santen Pharmaceutical Co. Ltd, Lupin, Allergan ple., LEO Pharma A/S, Aerie Pharmaceuticals Inc., Neurophth Therapeutics Inc, Stoke Therapeutics Inc, Bionure Farma S.L., Mallinckrodt Pharmaceuticals, Ixchel Pharma LLCGlobal Optic Atrophy Management Market Trends and Insights
Major companies operating in the optic atrophy management market are increasing their focus on conducting trials to develop targeted therapies for rare genetic disorders and gain a competitive edge. Targeted therapies for rare genetic disorders seek to tackle the fundamental genetic origin of the condition. This involves the transfer of genetic material into cells to rectify or substitute a faulty gene, addressing the root cause of the disease. For instance, in April 2023, Stoke Therapeutics, Inc., a US-based biotechnology company, received approval from the Medicines and Healthcare Products Regulatory Agency (MHRA) in the UK for its clinical trial application to initiate the Phase I/II OSPREY trial of STK-002. This trial aims to treat autosomal dominant optic atrophy (ADOA), which is the most prevalent inherited optic nerve disorder causing progressive and irreversible vision loss in both eyes, typically beginning in the first decade of life. Notably, there are currently no approved treatments for this condition, and STK-002 therapy is expected to decelerate or potentially stop the progression of vision loss in individuals affected by autosomal dominant optic atrophy (ADOA).What Are Latest Mergers And Acquisitions In The Optic Atrophy Management Market?
In January 2024, NAYA Biosciences, a US-based biotechnology company, acquired a clinical-stage gene therapy program for Leber’s Hereditary Optic Neuropathy (LHON) for an undisclosed amount. Through this acquisition, NAYA aims to strengthen its optic atrophy management capabilities by developing therapies that target optic nerve degeneration and preserve vision. Leber’s Hereditary Optic Neuropathy (LHON) is a US-based clinical-stage gene therapy program focused on treating a genetic disorder that causes optic nerve degeneration leading to vision loss.Regional Outlook
North America was the largest region in the optic atrophy management market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Optic Atrophy Management Market?
The optic atrophy management market consists of revenues earned by entities by providing services such as diagnosis consultations, visual rehabilitation services, and telemedicine services that help in determining causes and assessing the severity of optic nerve damage. The market value includes the value of related goods sold by the service provider or included within the service offering. The optic atrophy management market also includes sales of methylprednisolone, acetazolamide, antiviral medications, and immunosuppressants, which are used in the management of related conditions that lead to optic atrophy. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Optic Atrophy Management Market Report 2026?
The optic atrophy management market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the optic atrophy management industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Optic Atrophy Management Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $9.51 billion |
| Revenue Forecast In 2035 | $12.32 billion |
| Growth Rate | CAGR of 6.8% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Drug Type, Route Of Administration, Distribution Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Pfizer Inc., Johnson And Johnson, F. Hoffmann-La Roche AG, AbbVie Inc., Bayer AG, Novartis AG, Bristol-Myers Squibb Company, Sanofi SA, GlaxoSmithKline plc, Eli Lilly And Company, Boehringer Ingelheim International GmbH., Teva Pharmaceuticals, Viatris (Mylan N.V.), Astellas Pharma Inc, Bausch Health Companies Inc., Sumitomo Dainippon Pharma Co. Ltd, Bausch And Lomb Inc., Aurobindo Pharma, Cipla Ltd, Apotex Inc. , Zydus Cadila, Santen Pharmaceutical Co. Ltd, Lupin, Allergan ple., LEO Pharma A/S, Aerie Pharmaceuticals Inc., Neurophth Therapeutics Inc, Stoke Therapeutics Inc, Bionure Farma S.L., Mallinckrodt Pharmaceuticals, Ixchel Pharma LLC |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
