
Pancreatic Cancer Diagnostic Market Report 2026
Global Outlook – By Product (Instruments, Consumables, Services), By Treatment (Surgery, Chemotherapy, Radiation Therapy, Targeted Therapy), By Cancer Type (Exocrine, Adenocarcinoma, Squamous Cell Carcinoma, Adenosquamous Carcinoma, Colloid Carcinoma, Endocrine), By Test Type (Imaging Test, Biopsy, Blood Test, Genomic Test, Other Test Types), By End-User (Hospitals, Diagnostic Centers, Cancer Research Centers, Academic Institutes, Ambulatory Surgical Centers, Other End-Users) - Market Size, Trends, And Global Forecast 2026-2035
Pancreatic Cancer Diagnostic Market Overview
• Pancreatic Cancer Diagnostic market size has reached to $4.54 billion in 2025 • Expected to grow to $6.95 billion in 2030 at a compound annual growth rate (CAGR) of 9% • Growth Driver: Increase In Healthcare Spending Drives Growth In The Pancreatic Cancer Diagnostic Market • Market Trend: Prestige Biopharma Launches Groundbreaking Pauf-Detecting Diagnostic Kit For Early Detection Of Pancreatic Cancer • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Pancreatic Cancer Diagnostic Market?
Pancreatic cancer diagnostic refers to the procedures and methods for identifying, classifying, and staging pancreatic cancer to determine the condition, determining the severity of the disease, and directing treatment choices. It is commonly used to treat adenocarcinoma, squamous cell carcinoma, and insulinoma. The main types of pancreatic cancer diagnostic products are instruments, consumables, and services. An instrument refers to a tool or gadget that is used to carry out specified tasks or achieve desired results during a procedure, such as changing biological tissue or enabling access to examine it. Several types of treatment include surgery, chemotherapy, radiation therapy, and targeted therapy to treat various types of cancer, such as exocrine, adenocarcinoma, squamous cell carcinoma, adenosquamous carcinoma, colloid carcinoma, and endocrine. Various types of tests are included, such as imaging tests, biopsies, blood tests, genomic tests, and others, which are used by several end-users, including hospitals, diagnostic centers, cancer research centers, academic institutes, ambulatory surgical centers, and others.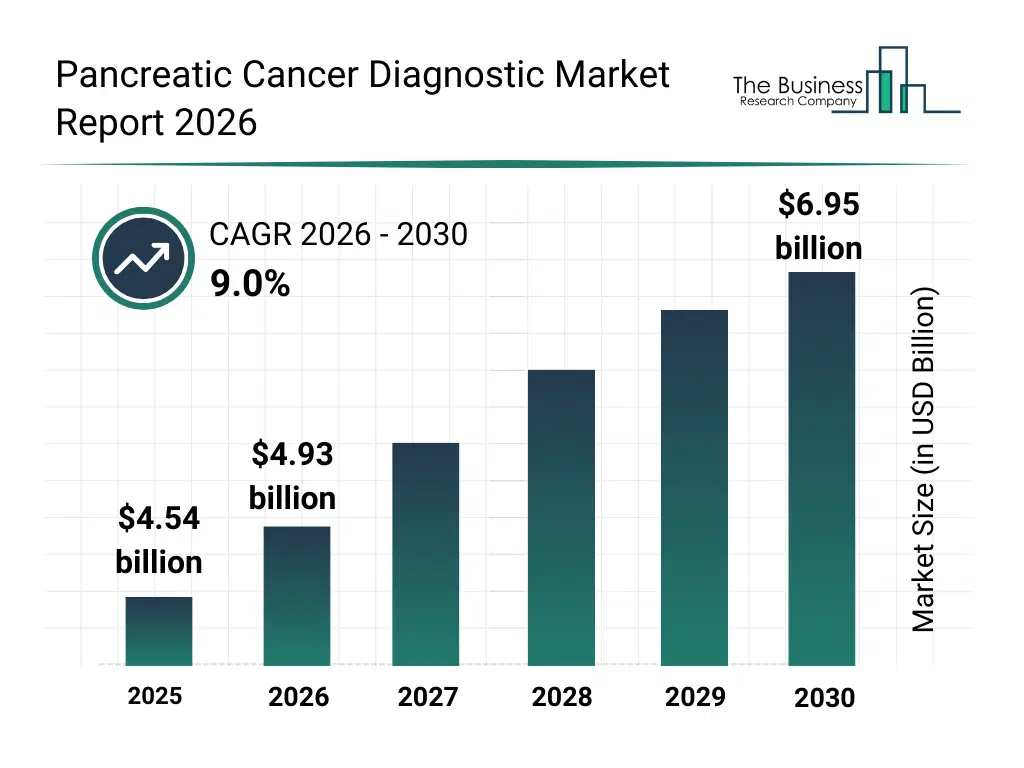
What Is The Pancreatic Cancer Diagnostic Market Size and Share 2026?
The pancreatic cancer diagnostic market size has grown strongly in recent years. It will grow from $4.54 billion in 2025 to $4.93 billion in 2026 at a compound annual growth rate (CAGR) of 8.6%. The growth in the historic period can be attributed to limited availability of advanced diagnostic tools, reliance on conventional imaging and biopsy, low early detection rates, fragmented diagnostic services, hospital-centric diagnostic approach.What Is The Pancreatic Cancer Diagnostic Market Growth Forecast?
The pancreatic cancer diagnostic market size is expected to see strong growth in the next few years. It will grow to $6.95 billion in 2030 at a compound annual growth rate (CAGR) of 9.0%. The growth in the forecast period can be attributed to increasing use of genomic and biomarker-based tests, adoption of ai and machine learning in diagnostics, growth in minimally invasive liquid biopsy techniques, expansion of diagnostic services in hospitals and cancer centers, rise in integrated laboratory and consulting services. Major trends in the forecast period include adoption of advanced genomic testing for pancreatic cancer, integration of ai-powered diagnostic imaging solutions, rise in liquid biopsy and minimally invasive tests, growth in precision medicine diagnostics, expansion of hospital and diagnostic center services.Global Pancreatic Cancer Diagnostic Market Segmentation
1) By Product: Instruments, Consumables, Services 2) By Treatment: Surgery, Chemotherapy, Radiation Therapy, Targeted Therapy 3) By Cancer Type: Exocrine, Adenocarcinoma, Squamous Cell Carcinoma, Adenosquamous Carcinoma, Colloid Carcinoma, Endocrine 4) By Test Type: Imaging Test, Biopsy, Blood Test, Genomic Test, Other Test Types 5) By End-User: Hospitals, Diagnostic Centers, Cancer Research Centers, Academic Institutes, Ambulatory Surgical Centers, Other End-Users Subsegments: 1) By Instruments: Imaging Systems, Biopsy Devices, Endoscopy Equipment 2) By Consumables: Reagents, Biopsy Needles, Assay Kits 3) By Services: Diagnostic Testing Services, Laboratory Services, Consulting ServicesWhat Is The Driver Of The Pancreatic Cancer Diagnostic Market?
The increase in healthcare spending is driving the growth of the pancreatic cancer diagnostic market going forward. Healthcare spending refers to the total amount of financial resources allocated to the healthcare sector within a specific region, country, or organization. Healthcare spending allocates resources to pancreatic cancer diagnostics to facilitate early detection, accurate staging, and timely intervention, aiming to improve patient outcomes and survival rates. For instance, in November 2023, according to the Canadian Institute for Health Information, a Canada-based institute for health information, in 2023, total health spending in Canada is projected to reach $344 billion, equating to $8,740 per person. This marks a 2.8% increase compared to the previous year, which saw only a 1.5% growth in health spending in 2022, indicating a notable rebound in healthcare investment. Therefore, an increase in healthcare spending is driving the growth of the pancreatic cancer diagnostic industry going forward.Key Players In The Global Pancreatic Cancer Diagnostic Market
Major companies operating in the pancreatic cancer diagnostic market are Hitachi Medical Corporation, F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories Inc., Danaher Corporation, Siemens Healthcare GmbH, FUJIFILM Corporation, Becton Dickinson and Company, Koninklijke Philips N.V., GE Healthcare Bio Sciences AB, Laboratory Corporation of America Holdings, Agilent Technologies Inc., Illumina Inc., Qiagen Inc., Myriad Genetics Inc., Canon Medical Systems Corporation, Biological Dynamics Inc., Berg LLC, Abcodia Ltd., Acobiom, Amplified Sciences LLC, Avant Diagnostics Inc., Axim Biotechnologies Inc., Alaunus Biosciences Inc., Advanced Marker Discovery SL, Anixa Diagnostics CorporationGlobal Pancreatic Cancer Diagnostic Market Trends and Insights
Major companies operating in the pancreatic cancer diagnostic market are focusing on developing innovative solutions, such as biomarker-based diagnostic kits, to meet the rising demand for early detection, improved diagnostic accuracy, and better patient survival outcomes. Biomarker-driven diagnostics identify tumor-associated proteins or genetic markers in blood or tissue samples, offering a more sensitive and specific approach compared with conventional imaging-led diagnosis, which often detects pancreatic cancer only at advanced stages. For instance, in March 2023, Prestige Biopharma Limited, a Singapore-based biopharmaceutical company, launched the first-in-class PAUF-detecting diagnostic kit, a powerful instrument for the early detection of pancreatic cancer. PAUF (Pancreatic Adenocarcinoma Upregulated Factor) is a unique tumor-specific biomarker associated with early metastasis and overexpression in about 80% of pancreatic cancer cases. The innovative diagnostic technique has high sensitivity and specificity for identifying PAUF. Preliminary studies showed that the expression level of PAUF in the blood plasma of pancreatic cancer patients is almost 1.9 times higher than that of healthy individuals, according to Prestige Biopharma's IDC (Innovative Discovery Center).What Are Latest Mergers And Acquisitions In The Pancreatic Cancer Diagnostic Market?
In January 2024, Bristol-Myers Squibb (BMS), a US-based biopharmaceutical company, acquired Mirati Therapeutics, Inc. for $4.8 billion. This strategic acquisition enhances BMS's portfolio by integrating Mirati's innovative cancer treatment assets, particularly those targeting pancreatic cancer. Through this deal, BMS aims to leverage its global scale and resources to expedite the delivery of more effective treatments to cancer patients. Mirati Therapeutics, Inc. is a US-based biotech company focused on pancreatic cancer diagnosis.Regional Outlook
North America was the largest region in the pancreatic cancer diagnostic market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pancreatic Cancer Diagnostic Market?
The pancreatic cancer diagnostic market includes revenues earned by entities through computerized tomography (CT) scans, Magnetic resonance imaging (MRI), positron emission tomography (PET) scan, and ultrasound. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included. The pancreatic cancer diagnostic market consists of sales of tumor markers and drugs such as Abraxane, Afinitor, and Capecitabine. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pancreatic Cancer Diagnostic Market Report 2026?
The pancreatic cancer diagnostic market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pancreatic cancer diagnostic industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pancreatic Cancer Diagnostic Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $4.93 billion |
| Revenue Forecast In 2035 | $6.95 billion |
| Growth Rate | CAGR of 8.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Treatment, Cancer Type, Test Type, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Hitachi Medical Corporation, F. Hoffmann-La Roche AG, Thermo Fisher Scientific Inc., Abbott Laboratories Inc., Danaher Corporation, Siemens Healthcare GmbH, FUJIFILM Corporation, Becton Dickinson and Company, Koninklijke Philips N.V., GE Healthcare Bio Sciences AB, Laboratory Corporation of America Holdings, Agilent Technologies Inc., Illumina Inc., Qiagen Inc., Myriad Genetics Inc., Canon Medical Systems Corporation, Biological Dynamics Inc., Berg LLC, Abcodia Ltd., Acobiom, Amplified Sciences LLC, Avant Diagnostics Inc., Axim Biotechnologies Inc., Alaunus Biosciences Inc., Advanced Marker Discovery SL, Anixa Diagnostics Corporation |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
