
Pediatric Neuroblastoma Treatment Market Report 2026
Global Outlook – By Treatment Type (Immunotherapy, Chemotherapy, Radiation Therapy, Other Treatment Types), By Risk Group (Low Risk, Intermediate Risk, High Risk), By End User (Hospitals, Specialty Clinics, Other End Users) - Market Size, Trends, And Global Forecast 2026-2035
Pediatric Neuroblastoma Treatment Market Overview
• Pediatric Neuroblastoma Treatment market size has reached to $2.02 billion in 2025 • Expected to grow to $3.13 billion in 2030 at a compound annual growth rate (CAGR) of 9.2% • Growth Driver: Surging Pediatric Neuroblastoma Cases Propel Advancements In Treatment Market A Focus On Research, Innovation, And Early Detection • Market Trend: Antibody Technologies in Pediatric Neuroblastoma Treatment • North America was the largest region in 2025.What Is Covered Under Pediatric Neuroblastoma Treatment Market?
Pediatric neuroblastoma treatment refers to a kind of treatment for cancer or a tumor that affects the nervous system in children. Neuroblastoma is typically treated with a combination of surgery, chemotherapy, radiation therapy, immunotherapy, and targeted therapy. The main types of pediatric neuroblastoma treatments are immunotherapy, chemotherapy, radiation therapy, and others. Immunotherapy is a form of medical treatment that uses the immune system's power to fight disease. It is used to treat several risk groups, such as low risk, intermediate risk, and high risk, and is used by various end users including hospitals, specialty clinics, and others.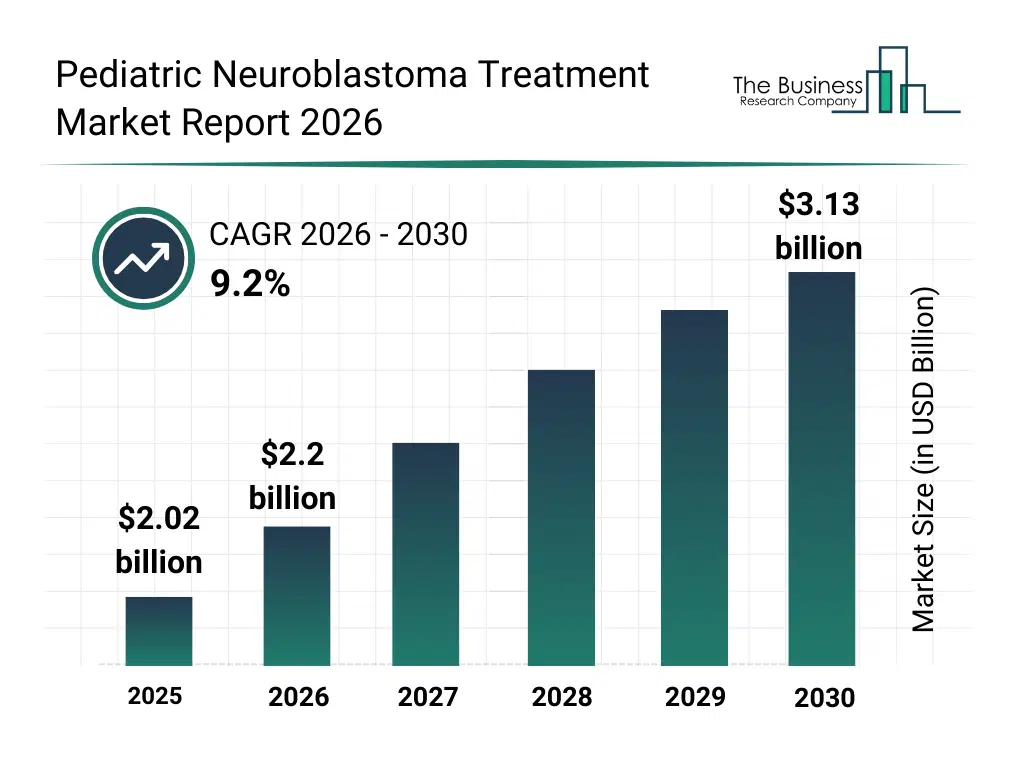
What Is The Pediatric Neuroblastoma Treatment Market Size and Share 2026?
The pediatric neuroblastoma treatment market size has grown strongly in recent years. It will grow from $2.02 billion in 2025 to $2.2 billion in 2026 at a compound annual growth rate (CAGR) of 9.0%. The growth in the historic period can be attributed to limited access to pediatric oncology drugs, low adoption of immunotherapy, reliance on conventional chemotherapy, insufficient specialized treatment centers, fragmented clinical data on neuroblastoma.What Is The Pediatric Neuroblastoma Treatment Market Growth Forecast?
The pediatric neuroblastoma treatment market size is expected to see strong growth in the next few years. It will grow to $3.13 billion in 2030 at a compound annual growth rate (CAGR) of 9.2%. The growth in the forecast period can be attributed to growing r&d in targeted therapies, increasing use of car t-cell and monoclonal antibody treatments, rising adoption of personalized medicine approaches, increasing investment in pediatric oncology infrastructure, expanding awareness programs for early detection. Major trends in the forecast period include increasing use of ai in pediatric oncology diagnostics, growing adoption of genomics-based targeted therapies, rising integration of cloud and big data for patient management, increasing use of iot-enabled monitoring in treatment protocols, rising implementation of smart manufacturing in drug production.Global Pediatric Neuroblastoma Treatment Market Segmentation
1) By Treatment Type: Immunotherapy, Chemotherapy, Radiation Therapy, Other Treatment Types 2) By Risk Group: Low Risk, Intermediate Risk, High Risk 3) By End User: Hospitals, Specialty Clinics, Other End Users Subsegments: 1) By Immunotherapy: Monoclonal Antibodies, Immune Checkpoint Inhibitors, CAR T-Cell Therapy 2) By Chemotherapy: Alkylating Agents, Antimetabolites, Topoisomerase Inhibitors 3) By Radiation Therapy: External Beam Radiation Therapy (EBRT), Radioisotope Therapy 4) By Other Treatment Types: Surgery, Stem Cell Transplantation, Targeted TherapyWhat Is The Driver Of The Pediatric Neuroblastoma Treatment Market?
The rising number of new cases of pediatric neuroblastoma is expected to propel the growth of the pediatric neuroblastoma treatment market going forward. The rising prevalence of neuroblastoma is increasing the demand for pediatric neuroblastoma treatment alternatives. As a result, pharmaceutical corporations and other stakeholders have boosted their investment in research and development, promoting innovation in the industry. Furthermore, the availability of new diagnostic technology and screening procedures has resulted in earlier neuroblastoma discovery, allowing for earlier treatments and better outcomes. For instance, in March 2023, according to an article published by the American Society of Clinical Oncology (ASCO), a US-based organization for oncology professionals and physicians, neuroblastoma affects approximately 700 to 800 youngsters in the United States each year. In the United States, neuroblastoma accounts for 6% of all childhood malignancies. Neuroblastoma affects around 90% of children under the age of five. Therefore, the rising number of new cases of pediatric neuroblastoma is driving the growth of the pediatric neuroblastoma treatment industry.Key Players In The Global Pediatric Neuroblastoma Treatment Market
Major companies operating in the pediatric neuroblastoma treatment market are United Therapeutics Corporation, APEIRON Biologics AG, Cell Ectar Biosciences Inc., Pfizer Inc., MacroGenics Inc., Bayer AG, Provectus Biopharmaceuticals Inc., Y-mAbs Therapeutics Inc., Amgen Inc., Eli Lilly and Company, F. Hoffmann-La Roche AG, Clarity Pharmaceuticals, CureSearch for Children's Cancer, Novartis AG, Johnson & Johnson Services Inc., AbbVie Inc., Merck & Co. Inc., Bristol-Myers Squibb Company, Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company Limited, Eisai Co. Ltd.Global Pediatric Neuroblastoma Treatment Market Trends and Insights
Major companies operating in the pediatric neuroblastoma treatment market are developing technologies such as antibody technologies to enhance targeted therapy efficacy and improve patient outcomes. Antibody technologies encompass various methods and tools for developing, producing, and utilizing antibodies for diagnostics, therapeutics, and research applications. For instance, in August 2023, Renaissance Pharma Ltd, a UK-based clinical-stage biopharmaceutical company, launched Hu14.18K322A. Hu14.18K322A targets GD2, a disialoganglioside present on the surface of neuroblastoma cells. By binding to these cells, it recruits immune effector cells to attack and kill the tumor cells, potentially improving treatment outcomes for patients with high-risk neuroblastoma.What Are Latest Mergers And Acquisitions In The Pediatric Neuroblastoma Treatment Market?
In September 2025, Serb Pharmaceuticals, a U.S.-based biopharmaceutical company focused on rare and oncology diseases, acquired Y-mAbs Therapeutics, Inc. for an undisclosed amount. With this acquisition, Serb aims to expand its pediatric oncology portfolio, gain access to Y-mAbs’ advanced neuroblastoma therapy, and accelerate commercial rollout of DANYELZA in high-risk pediatric neuroblastoma. Y-mAbs Therapeutics is a U.S.-based biopharma company specializing in antibody-based cancer therapies, including its lead anti-GD2 monoclonal antibody DANYELZA for relapsed or refractory high-risk pediatric neuroblastoma.Regional Outlook
North America was the largest region in the pediatric neuroblastoma treatment market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Pediatric Neuroblastoma Treatment Market?
The pediatric neuroblastoma treatment market consists of revenues earned by entities by providing pediatric neuroblastoma treatment services such as stem cell therapy, supportive care and blood transfusions. The market value includes the value of related goods sold by the service provider or included within the service offering. The pediatric neuroblastoma treatment market also includes sales of pain management medications, chemotherapy drugs and immunotherapy agents which are used in providing pediatric neuroblastoma treatment services. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Pediatric Neuroblastoma Treatment Market Report 2026?
The pediatric neuroblastoma treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the pediatric neuroblastoma treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Pediatric Neuroblastoma Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $2.2 billion |
| Revenue Forecast In 2035 | $3.13 billion |
| Growth Rate | CAGR of 9.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Treatment Type, Risk Group, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | United Therapeutics Corporation, APEIRON Biologics AG, Cell Ectar Biosciences Inc., Pfizer Inc., MacroGenics Inc., Bayer AG, Provectus Biopharmaceuticals Inc., Y-mAbs Therapeutics Inc., Amgen Inc., Eli Lilly and Company, F. Hoffmann-La Roche AG, Clarity Pharmaceuticals, CureSearch for Children's Cancer, Novartis AG, Johnson & Johnson Services Inc., AbbVie Inc., Merck & Co. Inc., Bristol-Myers Squibb Company, Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company Limited, Eisai Co. Ltd. |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
