
Point-of-Care (POC) Coagulation Testing Market Report 2026
Global Outlook – By Technology (Optical Technology, Mechanical Technology, Electrochemical Technology), By Distribution Channel (Direct Tenders, Retail), By End-User (Hospitals, Clinics, Home care) – Market Size, Trends, Strategies, and Forecast to 2035
Point-of-Care (POC) Coagulation Testing Market Overview
• Point-of-Care (POC) Coagulation Testing market size has reached to $1.25 billion in 2025 • Expected to grow to $1.52 billion in 2030 at a compound annual growth rate (CAGR) of 4.1% • Growth Driver: Cardiovascular Disease Rates Fuel Expansion Of POC Coagulation Testing Market • Market Trend: Increasing Focus On Innovating With Connected POC Coagulation Solutions In A Competitive Market • North America was the largest region in 2025.What Is Covered Under Point-of-Care (POC) Coagulation Testing Market?
Point-of-care (POC) coagulation testing refers to diagnostic tests that assess blood clotting function performed at the patient's bedside or near the site of care, rather than in a centralized laboratory. It is used to provide real-time or near-real-time results (within minutes), significantly faster than traditional lab tests, that allow healthcare professionals to make quicker treatment decisions and optimize anticoagulation therapy. The main types of point-of-care (POC) coagulation testing technologies include optical technology, mechanical technology, and electrochemical technology. Optical technology refers to the use of light for various applications in technology, computing, programming, the internet, and communications, involving the transmission, manipulation, and detection of light for data transfer, imaging, sensing, and other purposes. These are sold through several distribution channels, such as direct tenders and retail, to various end-users, including hospitals, clinics, and home care.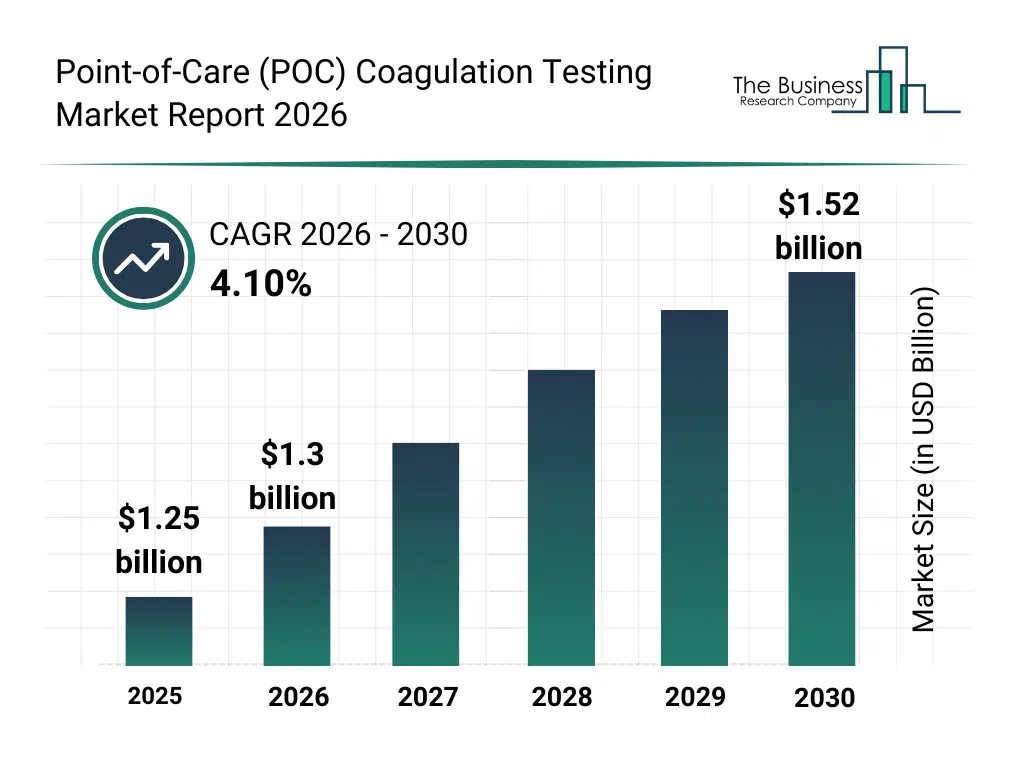
What Is The Point-of-Care (POC) Coagulation Testing Market Size and Share 2026?
The point-of-care (poc) coagulation testing market size has grown steadily in recent years. It will grow from $1.25 billion in 2025 to $1.3 billion in 2026 at a compound annual growth rate (CAGR) of 4.2%. The growth in the historic period can be attributed to rising prevalence of cardiovascular diseases requiring anticoagulation therapy, increasing demand for rapid coagulation results in operating rooms and icus, early clinical adoption of optical and mechanical poc testing platforms, expansion of coagulation monitoring in hospital and clinic settings, growing awareness of point-of-care diagnostics for treatment optimization.What Is The Point-of-Care (POC) Coagulation Testing Market Growth Forecast?
The point-of-care (poc) coagulation testing market size is expected to see steady growth in the next few years. It will grow to $1.52 billion in 2030 at a compound annual growth rate (CAGR) of 4.1%. The growth in the forecast period can be attributed to increasing shift toward decentralized and home-based coagulation monitoring, rising utilization of electrochemical detection technologies, growing procedure volumes in emergency and trauma care environments, expanding access to retail and direct tender distribution channels, rising focus on reducing turnaround time and improving therapy outcomes. Major trends in the forecast period include rising adoption of rapid bedside coagulation testing in critical and emergency care settings, increasing use of poc coagulation devices for anticoagulation therapy monitoring, growing preference for portable and handheld coagulation analyzers, expansion of coagulation testing utilization in home care and outpatient settings, increasing demand for real-time clinical decision support through near-patient testing.Global Point-of-Care (POC) Coagulation Testing Market Segmentation
1) By Technology: Optical Technology, Mechanical Technology, Electrochemical Technology 2) By Distribution Channel: Direct Tenders, Retail 3) By End-User: Hospitals, Clinics, Home care Subsegments: 1) By Optical Technology: Reflectance Photometry, Fluorescence Detection, Colorimetric Detection 2) By Mechanical Technology: Mechanical Oscillation, Ultrasonic Detection 3) By Electrochemical Technology: Amperometric Detection, Potentiometric Detection, Conductometric DetectionWhat Is The Driver Of The Point-of-Care (POC) Coagulation Testing Market?
The rising incidence of cardiovascular diseases is expected to propel the growth of the point-of-care (POC) coagulation testing market going forward. Cardiovascular diseases (CVDs) refer to a group of disorders of the heart and blood vessels, including conditions such as coronary heart disease, cerebrovascular disease, and peripheral arterial disease. Point-of-care (POC) coagulation testing is helpful in the treatment of cardiovascular diseases (CVDs) as it provides real-time information on coagulation status, enabling timely decision-making on coagulation interventions and reducing the risk of bleeding complications after cardiac surgery For instance, in October 2024, according to the Centers for Disease Control and Prevention, a US-based public health institute, in 2023, about 1 out of every 6 deaths from cardiovascular diseases was among adults younger than 65 years old, and a total of 919,032 people died from cardiovascular disease, representing 1 in every 3 deaths. Therefore, the rising incidence of cardiovascular diseases is driving the growth of the point-of-care (POC) coagulation testing industry.Key Players In The Global Point-of-Care (POC) Coagulation Testing Market
Major companies operating in the point-of-care (poc) coagulation testing market are Siemens Healthcare GmbH, Abbott Laboratories, F. Hoffmann-La Roche Ltd., Danaher Corporation, Sysmex Corporation, IDEXX Laboratories Inc., Quidel Corporation, Werfen SA, Nihon Kohden Corporation, Horiba Ltd., Hycel, Nova Biomedical Corporation, Haemonetics Corporation, Maccura Biotechnology Co. Ltd., Helena Laboratories Corporation, Abaxis Inc., ARKRAY Inc., Radiometer Medical ApS, HemoSonics LLC, iLine Microsystems SL, Corgenix Medical Corporation, Quotient Limited, Micropoint Bioscience Inc., Beijing Succeeder Technology Inc., EKF Diagnostics Holdings plcGlobal Point-of-Care (POC) Coagulation Testing Market Trends and Insights
Major companies operating in the point-of-care (POC) coagulation testing market are focused on innovating connected poc coagulation solutions, through strategic collaborations to gain a competitive edge in the market. Strategic collaboration is a formal agreement between two or more parties to work together toward achieving specific, predetermined goals that are mutually beneficial. For instance, in January 2023, Jana care, a US-based company developing biomarker assays for its proprietary digital health platform, collaborated with Roche Diagnostics, a Switzerland-based company that develop diagnostic tests, instruments and digital solutions with the power to transform healthcare for people. This collaboration aims to develop and distribute a point-of-care blood testing platform designed to screen for chronic kidney disease and heart failure. This innovative platform will enable patients to perform blood tests at home, with the results being reviewed remotely by clinicians.What Are Latest Mergers And Acquisitions In The Point-of-Care (POC) Coagulation Testing Market?
In December 2023, F. Hoffmann-La Roche AG, a Switzerland-based pharmaceutical company, acquired LumiraDx Ltd. for $350 million. The acquisition enables Roche's diagnostics to bolster its portfolio and enable more patient-centric healthcare with point-of-care solutions that span the entire patient journey. LumiraDx Ltd. is a UK-based provider of point-of-care (POC) coagulation testing.Regional Outlook
North America was the largest region in the point-of-care (POC) coagulation testing market in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Point-of-Care (POC) Coagulation Testing Market?
The point-of-care (POC) coagulation testing market consists of revenues earned by entities by providing coagulation testing services, on-site hemostasis testing, point-of-care diagnostic services, and rapid coagulation analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. The point-of-care (POC) coagulation testing market also includes sales of coagulation analyzers, test strips, prothrombin time (PT) monitors, D-dimer testing devices, and point-of-care coagulation testing kits. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Point-of-Care (POC) Coagulation Testing Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.3 billion |
| Revenue Forecast In 2035 | $1.52 billion |
| Growth Rate | CAGR of 4.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Distribution Channel, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Siemens Healthcare GmbH, Abbott Laboratories, F. Hoffmann-La Roche Ltd., Danaher Corporation, Sysmex Corporation, IDEXX Laboratories Inc., Quidel Corporation, Werfen SA, Nihon Kohden Corporation, Horiba Ltd., Hycel, Nova Biomedical Corporation, Haemonetics Corporation, Maccura Biotechnology Co. Ltd., Helena Laboratories Corporation, Abaxis Inc., ARKRAY Inc., Radiometer Medical ApS, HemoSonics LLC, iLine Microsystems SL, Corgenix Medical Corporation, Quotient Limited, Micropoint Bioscience Inc., Beijing Succeeder Technology Inc., EKF Diagnostics Holdings plc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
