
Preclinical CRO Market Report 2026
Global Outlook – By Service (Bioanalysis And DMPK Studies, Toxicology Testing, Compound Management, Chemistry, Safety Pharmacology, Others Services), By Type (Patient Derived Organoid (PDO) Model, Patient Derived Xenograft Model), By Animal Model (Small Animal Model, Large Animal Model), By Model System (In Vivo, In Vitro), By End User (Biopharmaceutical Companies, Government And Academic Institutes, Medical Device Companies, Other End-users) - Market Size, Trends, And Global Forecast 2026-2035
Preclinical CRO Market Overview
• Preclinical CRO market size has reached to $6.25 billion in 2025 • Expected to grow to $9.83 billion in 2030 at a compound annual growth rate (CAGR) of 9.5% • Growth Driver: Surging Demand For Preclinical Trials Drives Growth In The Preclinical CRO Market • Market Trend: Revolutionizing Preclinical Drug Discovery • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Preclinical CRO Market?
Preclinical CROs (contract research organizations) are support organizations that provide the knowledge in research and development needed to guide a drug candidate through animal testing and advance it to the clinical stage. A preclinical CRO supports new medical product producers in showing product safety and efficacy in living animals that the Food and Drug Administration (FDA) considers mirroring human anatomy most closely before entering clinical trials or being used for human care. The main services of preclinical CRO are bioanalysis and DMPK studies, toxicology testing, compound management, chemistry, safety pharmacology, and others. A small three-dimensional (3D) cell culture made from a patient's cancer cells is known as a patient-derived organoid (PDO), and it has undergone substantial investment and experimental validation to offer prospective therapeutic alternatives for the diagnosis of complex diseases. The various types of preclinical CRO including the patient-derived organoid (PDO) model and the patient-derived xenograft model are available for small animal and large animal models. The model systems In Vivo and In Vitro are used by biopharmaceutical companies, government and academic institutes, medical device companies, and others.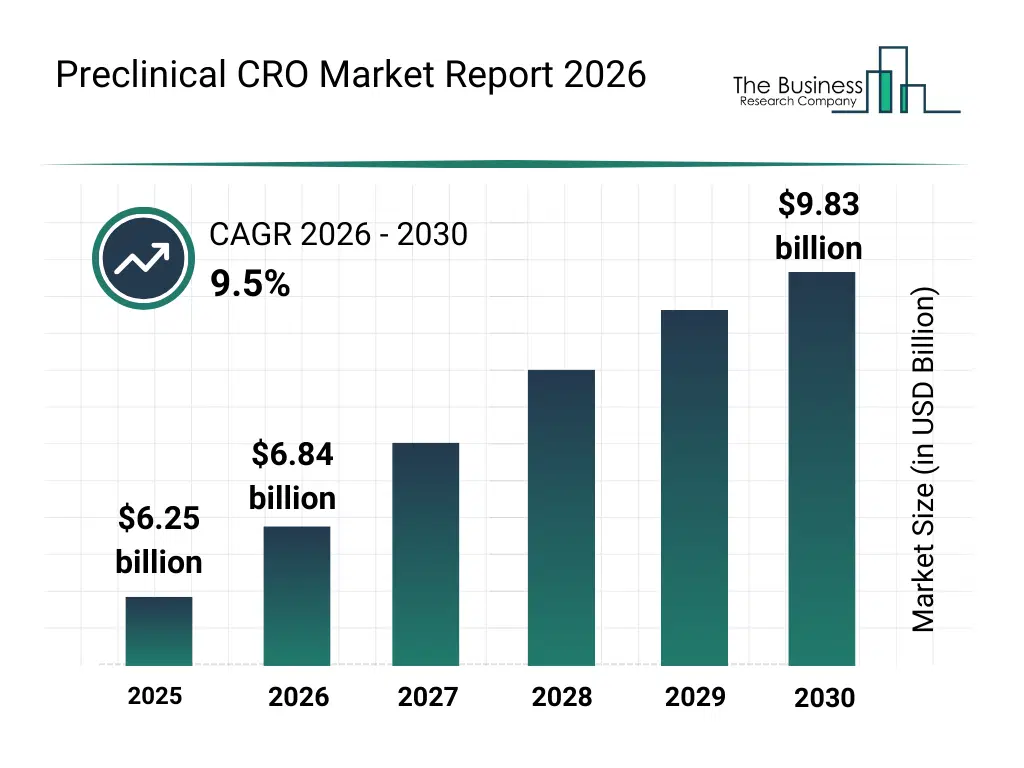
What Is The Preclinical CRO Market Size and Share 2026?
The preclinical cro market size has grown strongly in recent years. It will grow from $6.25 billion in 2025 to $6.84 billion in 2026 at a compound annual growth rate (CAGR) of 9.6%. The growth in the historic period can be attributed to increasing pharmaceutical R&D investments, rising complexity of drug development pipelines, growing reliance on animal testing for safety validation, expansion of biotechnology research activities, increased regulatory scrutiny on preclinical data.What Is The Preclinical CRO Market Growth Forecast?
The preclinical cro market size is expected to see strong growth in the next few years. It will grow to $9.83 billion in 2030 at a compound annual growth rate (CAGR) of 9.5%. The growth in the forecast period can be attributed to increasing demand for precision medicine research, rising focus on rare disease drug development, expansion of biologics and cell therapy pipelines, growing adoption of digital preclinical platforms, increasing partnerships between pharma companies and CROs. Major trends in the forecast period include increasing adoption of integrated preclinical study services, rising demand for advanced animal model expertise, growing outsourcing of early-stage drug development, expansion of regulatory-aligned toxicology capabilities, enhanced focus on data-driven study design.Global Preclinical CRO Market Segmentation
1) By Service: Bioanalysis And DMPK Studies, Toxicology Testing, Compound Management, Chemistry, Safety Pharmacology, Others Services 2) By Type: Patient Derived Organoid (PDO) Model, Patient Derived Xenograft Model 3) By Animal Model: Small Animal Model, Large Animal Model 4) By Model System: In Vivo, In Vitro 5) By End User: Biopharmaceutical Companies, Government And Academic Institutes, Medical Device Companies, Other End-users Subsegments: 1) By Bioanalysis And DMPK Studies: Pharmacokinetics Studies, Pharmacodynamics Studies, Drug Metabolism Studies 2) By Toxicology Testing: Acute Toxicity Testing, Chronic Toxicity Testing, Reproductive Toxicity Testing 3) By Compound Management: Sample Management, Inventory Management, Formulation Services 4) By Chemistry: Medicinal Chemistry, Analytical Chemistry, Synthetic Chemistry 5) By Safety Pharmacology: Cardiovascular Safety Studies, Central Nervous System Safety Studies, Respiratory Safety Studies 6) By Other Services: Regulatory Consulting, Project Management, Data Management ServicesWhat Is The Driver Of The Preclinical CRO Market?
The increasing demand for preclinical trials is expected to propel the growth of the preclinical CRO market going forward. Preclinical trials refer to any study conducted on medicine or treatment for a condition before human volunteers are used as test subjects. Preclinical studies often involve novel medical devices, prescription medications, and diagnostics, and its primary objectives are to establish a beginning, safe dose for first-in-human research and evaluate any potential toxicity of the product. Preclinical trials play a critical role in drug development, and their importance is increasing with the growing emphasis on the safety and efficacy of new drugs. For instance, in February 2024, according to IQVIA, a US-based company serving the combined industries of health information technology and clinical research reports, clinical development productivity rose in 2023, where a total of 69 novel active substances (NASs) were launched globally in 2023, 6 more than the prior year, and including 24 first-in-class launches in the U.S. Therefore, the increasing demand for preclinical trials is driving the growth of the preclinical CRO industry going forward.Key Players In The Global Preclinical CRO Market
Major companies operating in the preclinical cro market are Eurofins Scientific SE, WuXi AppTec Co Ltd, Medpace Holdings Inc, Charles River Laboratories International Inc, Laboratory Corporation Of America Holdings, Intertek Group plc, Crown Bioscience International, Cynbiose, AmplifyBio LLC, Vivotecnia SL, Altasciences Company Inc, Pharmaceutical Product Development LLC, Parexel International Corporation, Envigo RMS Holding Corporation, Veeda Clinical Research Ltd, ICON plc, Kunming Biomed International Ltd, PharmaLegacy Laboratories, NorthEast BioAnalytical Laboratories LLC, MDS Pharma Services, MPI Research, Toxikon Corporation, BioReliance Corporation, Covance Inc, Syngene International LimitedGlobal Preclinical CRO Market Trends and Insights
Major companies operating in the preclinical CRO market are adopting new technologies to sustain their position such as a disease biology knowledge graph to enhance target identification and drug discovery in pharmaceutical research. The disease biology knowledge graph is a structured, comprehensive representation of the relationships between genes, proteins, pathways, and disease phenotypes, designed to integrate diverse biological data. It enables researchers to systematically explore disease mechanisms, identify potential therapeutic targets, and accelerate drug discovery by providing a holistic map of disease biology. For instance, in February 2023, BenchSci, a Canada-based AI solutions company for preclinical R&D, launched ASCEND, an end-to-end SaaS (software as a service) platform and for preclinical drug discovery. By gaining biological insights into the fundamental underpinnings of disease, this method seeks to speed up preclinical phase drug development pipelines. ASCEND extracts experimental data from safe internal and public external sources using BenchSci's machine-learning technology. The platform compares experimental results using curated ontology datasets. As a result, the software can produce a map of the biological processes underpinning various diseases that are based on evidence.What Are Latest Mergers And Acquisitions In The Preclinical CRO Market?
In September 2024, PharmaLegacy Laboratories, a US-based preclinical drug development CRO, acquired BTS Research for an undisclosed amount. Through this acquisition, PharmaLegacy aims to expand its in vitro and in vivo preclinical services, increase study capacity across North America, and strengthen its global service offerings. BTS Research is a US-based preclinical CRO providing GLP and non-GLP biological services to pharmaceutical, biotech, medical-device, and diagnostic clients.Regional Outlook
North America was the largest region in the preclinical CRO market in 2025. Asia-Pacific is expected to be the fastest-growing region in the global preclinical CRO market during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Preclinical CRO Market?
The preclinical CRO market includes revenues earned by entities through project management, information gathering, medical evaluations, maximum dose research, adherence to regulations, efficacy and safety reporting, and quality analysis. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Preclinical CRO Market Report 2026?
The preclinical cro market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the preclinical cro industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Preclinical CRO Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $6.84 billion |
| Revenue Forecast In 2035 | $9.83 billion |
| Growth Rate | CAGR of 9.6% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Service, Type, Animal Model, Model System, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Eurofins Scientific SE, WuXi AppTec Co Ltd, Medpace Holdings Inc, Charles River Laboratories International Inc, Laboratory Corporation Of America Holdings, Intertek Group plc, Crown Bioscience International, Cynbiose, AmplifyBio LLC, Vivotecnia SL, Altasciences Company Inc, Pharmaceutical Product Development LLC, Parexel International Corporation, Envigo RMS Holding Corporation, Veeda Clinical Research Ltd, ICON plc, Kunming Biomed International Ltd, PharmaLegacy Laboratories, NorthEast BioAnalytical Laboratories LLC, MDS Pharma Services, MPI Research, Toxikon Corporation, BioReliance Corporation, Covance Inc, Syngene International Limited |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
