
Tumor Ablation Therapy Devices Market Report 2026
Global Outlook – By Technology (Radiofrequency Ablation, Microwave Ablation, Cryoablation, Irreversible Electroporation Ablation, Other Technologies), By Treatment (Surgical, Laparoscopic, Percutaneous), By Application (Kidney Cancer, Liver Cancer, Breast Cancer, Lung Cancer, Prostate Cancer, Other Applications), By End User (Hospitals, Oncology Clinics, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Tumor Ablation Therapy Devices Market Overview
• Tumor Ablation Therapy Devices market size has reached to $0.82 billion in 2025 • Expected to grow to $1.61 billion in 2030 at a compound annual growth rate (CAGR) of 14.2% • Growth Driver: Increasing Prevalence Of Cancer Drives Tumor Ablation Therapy Device Market Growth • Market Trend: Increasing Focus On Developing Minimally Invasive Therapies To Gain A Competitive Edge • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Tumor Ablation Therapy Devices Market?
Tumor ablation therapy devices are minimally invasive devices used in the treatment of tumors, commonly of kidney, liver, and lung. These devices work by employing various techniques to destroy cancerous cells while preserving surrounding healthy tissue. The main technologies in the tumor ablation therapy devices are radiofrequency ablation, microwave ablation, cryoablation, irreversible electroporation ablation, and others. Irreversible electroporation (IRE) in tumor ablation therapy is a new tissue ablation method in which micro to millisecond electrical pulses are supplied to undesirable tissue to produce cell necrosis by irreversible cell membrane permeabilization. The treatments included are surgical, laparoscopic, and percutaneous that are used in various applications such as kidney cancer, liver cancer, breast cancer, lung cancer, prostate cancer, and others. The various end-users of tumor ablation therapy devices are hospitals, oncology clinics, and others.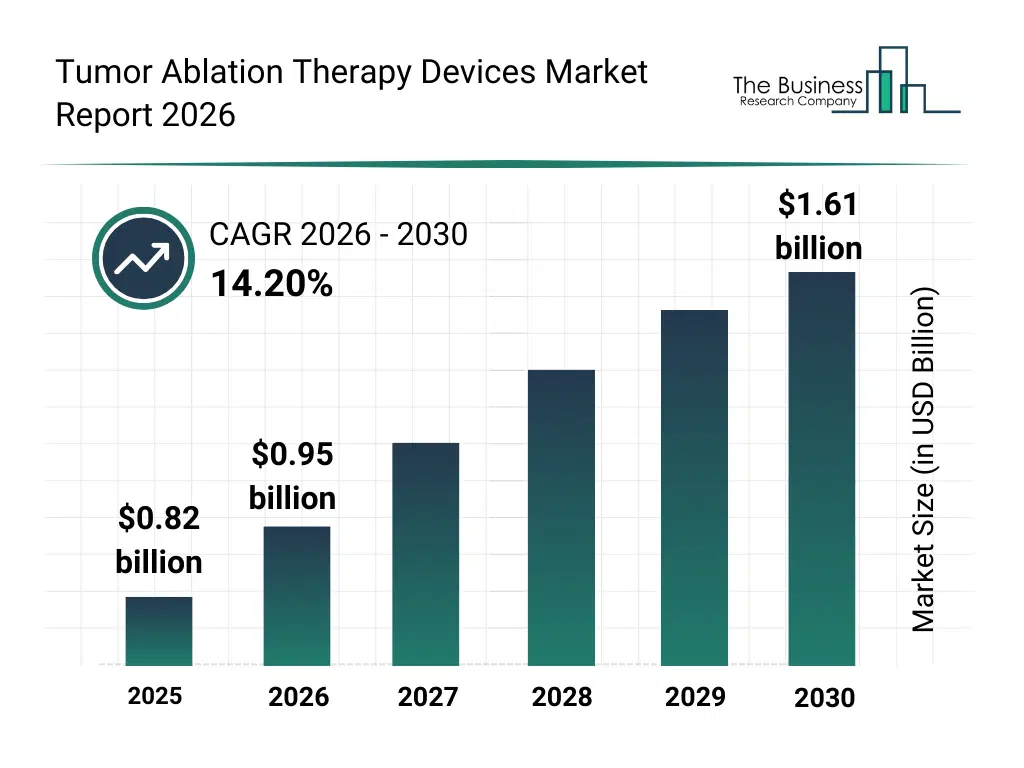
What Is The Tumor Ablation Therapy Devices Market Size and Share 2026?
The tumor ablation therapy devices market size has grown rapidly in recent years. It will grow from $0.82 billion in 2025 to $0.95 billion in 2026 at a compound annual growth rate (CAGR) of 15.0%. The growth in the historic period can be attributed to increasing cancer incidence rates, expansion of oncology treatment centers, rising adoption of minimally invasive therapies, availability of advanced imaging technologies, growing preference for organ-preserving treatments.What Is The Tumor Ablation Therapy Devices Market Growth Forecast?
The tumor ablation therapy devices market size is expected to see rapid growth in the next few years. It will grow to $1.61 billion in 2030 at a compound annual growth rate (CAGR) of 14.2%. The growth in the forecast period can be attributed to increasing investments in precision oncology, rising demand for outpatient cancer treatments, expansion of advanced ablation technologies, growing integration of imaging and therapy systems, increasing focus on personalized cancer care. Major trends in the forecast period include increasing adoption of minimally invasive tumor ablation procedures, rising use of image-guided ablation systems, growing demand for organ-specific ablation technologies, expansion of percutaneous treatment approaches, enhanced focus on precision oncology.Global Tumor Ablation Therapy Devices Market Segmentation
1) By Technology: Radiofrequency Ablation, Microwave Ablation, Cryoablation, Irreversible Electroporation Ablation, Other Technologies 2) By Treatment: Surgical, Laparoscopic, Percutaneous 3) By Application: Kidney Cancer, Liver Cancer, Breast Cancer, Lung Cancer, Prostate Cancer, Other Applications 4) By End User: Hospitals, Oncology Clinics, Other End Users Subsegments: 1) By Radiofrequency Ablation (RFA): Monopolar RFA Devices, Bipolar RFA Devices 2) By Microwave Ablation: High-frequency Microwave Devices, Low-frequency Microwave Devices 3) By Cryoablation: Liquid Nitrogen-based Devices, Argon Gas-based Devices 4) By Irreversible Electroporation Ablation (IRE): NanoKnife Ablation Systems, High-voltage Pulse Generators 5) By Other Technologies: Laser Ablation Devices, Ultrasound-guided High-intensity Focused Ultrasound (HIFU), Chemical Ablation TechniquesWhat Is The Driver Of The Tumor Ablation Therapy Devices Market?
The increasing prevalence of cancer is a key factor driving the growth of the tumor ablation therapy devices market. Cancer is considered to be the second major cause of death, with around 1 in 6 deaths due to cancer worldwide. The increasing prevalence of cancer is driven by aging populations, as older individuals have a higher risk of developing various types of cancer over their lifetime. Tumor ablation is a minimally invasive procedure widely used in the treatment of lung, liver, kidney, and bone tumors. For instance, in 2023, according to the American Cancer Society Inc., a US-based non-profit health group, 1,958,310 new cancer cases are expected to be diagnosed in the US, an increase compared to more than 1,918,030 new cancer cases in 2022. The procedure is an effective method for patients who have failed chemotherapy or radiotherapy, thereby supporting the tumor ablation therapy devices industry growth.Key Players In The Global Tumor Ablation Therapy Devices Market
Major companies operating in the tumor ablation therapy devices market are Galil Medical Inc., Misonix Inc., HealthTronics Inc., AngioDynamics Inc., Boston Scientific Corporation, Medtronic plc, EDAP TMS S.A., NeuWave Medical Inc., BVM Medical Limited, Erbe Elektromedizin GmbH, Theraclion, INTIO Inc., Olympus Corporation, Stryker Corporation, Merit Medical Systems Inc., Insightec Ltd., Terumo Corporation, Monteris Medical Corporation, IceCure Medical Ltd., Profound Medical Inc., Alpinion Medical Systems Co. Ltd., Sonacare Medical, Hologic Inc., Endocare Inc., Bovie Medical Corporation,Global Tumor Ablation Therapy Devices Market Trends and Insights
Major companies operating in the tumor ablation therapy devices market are focused on developing minimally invasive therapy to gain a competitive edge in the market. Minimally invasive therapy is utilized for tumor ablation, offering precise and effective treatment with minimal disruption to surrounding tissues. For instance, in August 2023, Francis Medical Inc., a US-based provider of minimally invasive treatment solutions for prostate, kidney, and bladder cancer, the U.S. Food and Drug Administration (FDA) The U.S. Food and Drug Administration (FDA) has approved Breakthrough Device Designation to its Vanquish minimally invasive water vapor ablation therapy. This aims to accelerate the device's development and streamline its regulatory evaluation, potentially resulting in quicker access for patients. Vanquish utilizes the thermal energy contained in a small amount of sterile water to administer precise treatments to cancerous tissue through a straightforward transurethral procedure. The treatment is engineered to destroy cancer cells while safeguarding nearby structures, thus diminishing the chances of severe side effects that often accompany alternative prostate cancer therapies.What Are Latest Mergers And Acquisitions In The Tumor Ablation Therapy Devices Market?
In October 2024, ExcelFin Acquisition Corp, a US-based publicly traded special purpose acquisition company, merged with Betters Medical Investment Holdings Limited (Baird Medical) for an undisclosed amount. With this merger, ExcelFin aims to take Baird Medical public and support its expansion strategy, enabling the company to scale its microwave ablation (MWA) technology portfolio and accelerate its entry into the U.S. and international markets. Baird Medical Investment Holdings Limited is a China-based medical device developer specializing in microwave ablation systems for minimally invasive tumor treatment.Regional Outlook
North America was the largest region in the tumor ablation therapy devices market in 2025. Asia-Pacific was the second largest region in the tumor ablation therapy devices market. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Tumor Ablation Therapy Devices Market?
The tumor ablation therapy devices market consists of sales of radiofrequency ablation, microwave ablation, cryoablation, irreversible electroporation ablation, and others that are used for tumor ablation therapies. Values in this market are factory gate values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Tumor Ablation Therapy Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $0.95 billion |
| Revenue Forecast In 2035 | $1.61 billion |
| Growth Rate | CAGR of 15.0% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Technology, Treatment, Application, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Galil Medical Inc., Misonix Inc., HealthTronics Inc., AngioDynamics Inc., Boston Scientific Corporation, Medtronic plc, EDAP TMS S.A., NeuWave Medical Inc., BVM Medical Limited, Erbe Elektromedizin GmbH, Theraclion, INTIO Inc., Olympus Corporation, Stryker Corporation, Merit Medical Systems Inc., Insightec Ltd., Terumo Corporation, Monteris Medical Corporation, IceCure Medical Ltd., Profound Medical Inc., Alpinion Medical Systems Co. Ltd., Sonacare Medical, Hologic Inc., Endocare Inc., Bovie Medical Corporation, |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
