
Hepatorenal Syndrome Treatment Market Report 2026
Global Outlook – By Type (Type 1 Hepatorenal Syndrome, Type 2 Hepatorenal Syndrome), By Treatment (Therapeutics, Surgical Treatment), By End-User (Hospitals And Clinics, Ambulatory Surgical Centers, Academic And Research Institutes, Other End-Users) – Market Size, Trends, Strategies, and Forecast to 2035
Hepatorenal Syndrome Treatment Market Overview
• Hepatorenal Syndrome Treatment market size has reached to $15.17 billion in 2025 • Expected to grow to $21.12 billion in 2030 at a compound annual growth rate (CAGR) of 7% • Growth Driver: Accelerated Drug Designations Catalyzing Growth In The Hepatorenal Syndrome Treatment Market • Market Trend: Advanced Renal-Support Therapy Shows Promising Outcomes In Severe Liver-Related Kidney Failure • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Hepatorenal Syndrome Treatment Market?
Hepatorenal syndrome (HRS) is a multiorgan disorder characterized by acute kidney injury in patients with severe liver disease. Patients with this syndrome display signs and symptoms of liver failure in addition to decreased urination when they become oliguric. The main types of hepatorenal syndrome treatment include type 1 hepatorenal syndrome and type 2 hepatorenal syndrome. Type 2 hepatorenal syndrome is distinguished by a moderate and steady decline in glomerular filtration rate, and it frequently affects people whose liver function is still mostly intact. They are treated by providing therapeutics and surgical treatment for use in various end-users such as hospitals and clinics, ambulatory surgical centers, academic and research institutes and others.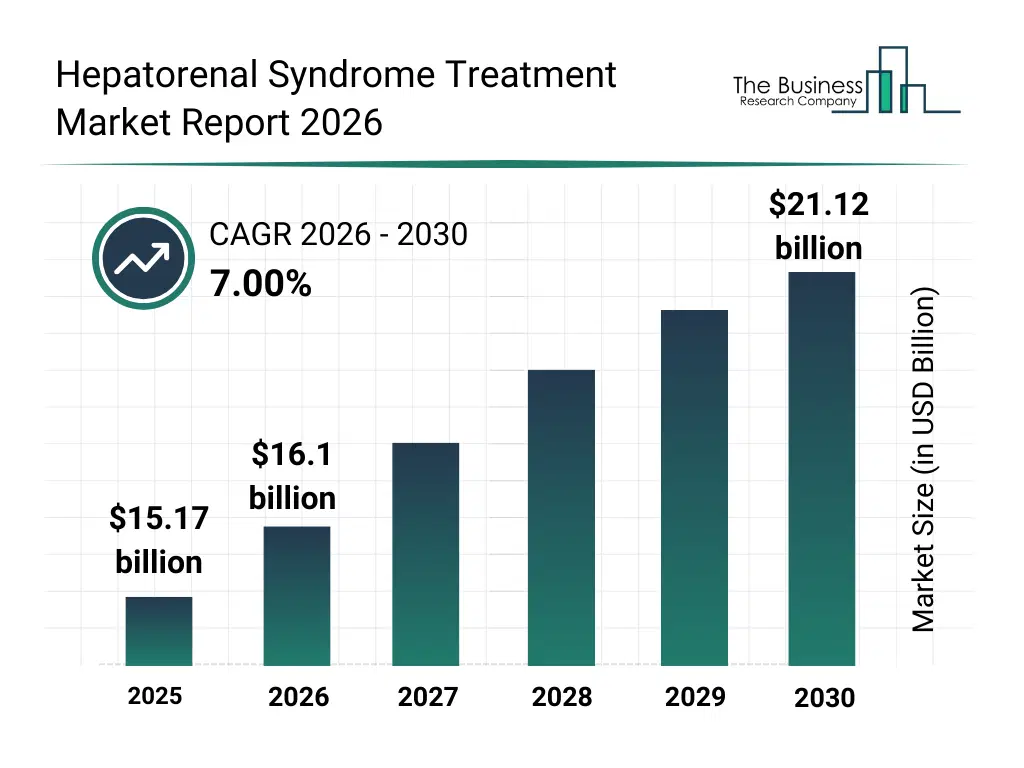
What Is The Hepatorenal Syndrome Treatment Market Size and Share 2026?
The hepatorenal syndrome treatment market size has grown strongly in recent years. It will grow from $15.17 billion in 2025 to $16.1 billion in 2026 at a compound annual growth rate (CAGR) of 6.1%. The growth in the historic period can be attributed to limited awareness of hrs, lack of standardized treatment protocols, high mortality associated with liver failure, dependency on conventional dialysis methods, restricted access to advanced therapeutics.What Is The Hepatorenal Syndrome Treatment Market Growth Forecast?
The hepatorenal syndrome treatment market size is expected to see strong growth in the next few years. It will grow to $21.12 billion in 2030 at a compound annual growth rate (CAGR) of 7.0%. The growth in the forecast period can be attributed to advancements in targeted therapies, adoption of precision medicine approaches, expansion of healthcare infrastructure, integration of ai and digital tools in patient monitoring, increased investment in novel drug development. Major trends in the forecast period include increasing prevalence of chronic liver diseases, advancements in renal replacement therapies, personalized treatment approaches for hrs, growth in hospital and clinical infrastructure, rising focus on early diagnosis and monitoring.Global Hepatorenal Syndrome Treatment Market Segmentation
1) By Type: Type 1 Hepatorenal Syndrome, Type 2 Hepatorenal Syndrome 2) By Treatment: Therapeutics, Surgical Treatment 3) By End-User: Hospitals And Clinics, Ambulatory Surgical Centers, Academic And Research Institutes, Other End-Users Subsegments: 1) By Type 1 Hepatorenal Syndrome: Intravenous Albumin, Vasopressors, Dialysis, Liver Transplantation 2) By Type 2 Hepatorenal Syndrome: Albumin Infusion, Midodrine, Octreotide, Liver TransplantationWhat Are The Drivers Of The Hepatorenal Syndrome Treatment Market?
The rise in fast-track and novel drug designations is expected to propel the growth of the hepatorenal syndrome treatment market going forward. Fast track and novel drug designations are regulatory pathways that expedite the development, review, and approval of drugs for serious conditions with unmet medical needs, aiming to bring innovative therapies to patients more quickly. The rise in fast-track and novel drug designations aims to expedite the development of treatments for hepatorenal syndrome by addressing the urgent need for effective therapies, potentially improving patient outcomes, and addressing unmet medical needs in this condition. For instance, in January 2024, according to the New Drug Therapy Approvals 2023 report published by the Center for Drug Evaluation and Research (CDER), a US-based branch of the U.S. Food and Drug Administration (FDA), the CDER approved 55 novel drugs in 2023, with 20 (36%) identified as first-in-class, compared to 37 novel drug approvals in 2022. Therefore, the rise in fast-track and novel drug designations is driving the growth of the hepatorenal syndrome treatment industry. An increase in healthcare spending is expected to propel the growth of the hepatorenal syndrome treatment market going forward. Healthcare spending is the total amount spent on healthcare throughout all sectors of an economy, including hospitals, home health services, prescription pharmaceuticals, nursing homes, and individual healthcare. As healthcare spending rises, more funding may be given to research and development of novel, more potent HRS treatments. Increased spending may also result in greater availability of tools for HRS screening and early detection, which could lead to earlier treatment and better results. For instance, in April 2025, according to the Office for National Statistics (ONS), a UK-based independent producer of official statistics, total healthcare expenditure rose by 6.5% in nominal terms and 2.4% in real terms between 2023 and 2024, while healthcare’s share of GDP held steady at 11.1% in 2024, matching the previous year. Therefore, the increase in healthcare spending is driving the growth of the hepatorenal syndrome treatment industry.Key Players In The Global Hepatorenal Syndrome Treatment Market
Major companies operating in the hepatorenal syndrome treatment market are BioVie Inc., Cumberland Pharmaceuticals Inc., Mallinckrodt Pharmaceuticals, Orphan Therapeutics LLC, Baxter International Inc., Novartis AG, Eli Lilly and Company, Johnson & Johnson, Boehringer Ingelheim GmbH, Bayer AG, Gilead Sciences Inc., Pfizer Inc., AstraZeneca PLC, AbbVie Inc., Amgen Inc., F Hoffmann-La Roche Ltd, Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company Limited, Teva Pharmaceutical Industries Ltd., Otsuka Pharmaceutical Co Ltd, Astellas Pharma Inc., Kyowa Kirin Co LtdGlobal Hepatorenal Syndrome Treatment Market Trends and Insights
Major companies operating in the hepatorenal syndrome treatment market are focusing on developing innovative advancements such as vasoconstrictive renal-support therapies to improve kidney function and clinical outcomes in patients experiencing rapid deterioration associated with advanced liver disease. A vasoconstrictive renal-support therapy is a pharmacological treatment designed to enhance renal perfusion and support kidney recovery by selectively constricting splanchnic blood vessels and stabilizing circulatory function. For instance, in June 2024, Mallinckrodt plc, an Ireland-based specialty pharmaceutical company, presented new clinical data on TERLIVAZ, a terlipressin injection approved for hepatorenal syndrome with rapid reduction in kidney function. The findings demonstrated favorable outcomes across several patient subgroups, including improved reversal rates of kidney dysfunction and reduced need for renal replacement therapy. The data further underscored the therapy’s relevance in addressing a life-threatening complication of advanced liver disease and its potential to inform earlier diagnosis and more effective clinical management.Regional Insights
North America was the largest region in the hepatorenal syndrome treatment market in 2025. Asia-Pacific is expected to be the fastest-growing region in the global hepatorenal syndrome treatment market during the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Hepatorenal Syndrome Treatment Market?
The hepatorenal syndrome treatment market includes revenues earned by providing imaging, blood, and urine tests to evaluate the patient's liver and kidney function. The market value includes the value of related goods sold by the service provider or included within the service offering. Only goods and services traded between entities or sold to end consumers are included.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Hepatorenal Syndrome Treatment Market Report 2026?
The hepatorenal syndrome treatment market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the hepatorenal syndrome treatment industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Hepatorenal Syndrome Treatment Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $16.1 billion |
| Revenue Forecast In 2035 | $21.12 billion |
| Growth Rate | CAGR of 6.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Treatment, End-User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | BioVie Inc., Cumberland Pharmaceuticals Inc., Mallinckrodt Pharmaceuticals, Orphan Therapeutics LLC, Baxter International Inc., Novartis AG, Eli Lilly and Company, Johnson & Johnson, Boehringer Ingelheim GmbH, Bayer AG, Gilead Sciences Inc., Pfizer Inc., AstraZeneca PLC, AbbVie Inc., Amgen Inc., F Hoffmann-La Roche Ltd, Takeda Pharmaceutical Company Limited, Daiichi Sankyo Company Limited, Teva Pharmaceutical Industries Ltd., Otsuka Pharmaceutical Co Ltd, Astellas Pharma Inc., Kyowa Kirin Co Ltd |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
