Neurovascular Embolization Devices Market Report 2026

Neurovascular Embolization Devices Market Report 2026
Global Outlook – By Product (Embolization Coils, Liquid Embolic Systems, Flow Diversion Stents, Aneurysm Clips, Other Products), By Treatment (Neurovascular Hemorrhage Management, Hemorrhagic Stroke Solutions), By Indication (Brain Aneurysms, Arteriovenous Malformation, Other Indications), By End User (Hospitals, Ambulatory Surgical Centers, Other End Users) – Market Size, Trends, Strategies, and Forecast to 2035
Neurovascular Embolization Devices Market Overview
• Neurovascular Embolization Devices market size has reached to $1.38 billion in 2025 • Expected to grow to $1.91 billion in 2030 at a compound annual growth rate (CAGR) of 6.8% • Growth Driver: Rising Hemorrhages Drive Neurovascular Embolization Devices Market Growth • Market Trend: Stryker Introduces Innovative Flow Diverter for Enhanced Aneurysm Treatment • North America was the largest region in 2025 and Asia-Pacific is the fastest growing region.What Is Covered Under Neurovascular Embolization Devices Market?
Neurovascular embolization devices are medical instruments used to treat abnormal brain or central nervous blood vessels by blocking or redirecting blood flow. These devices are employed in neuro-interventional procedures to manage conditions such as aneurysms, arteriovenous malformations (AVMs), and arteriovenous fistulas (AVFs) that may risk bleeding or other complications. The main products of neurovascular embolization devices included are embolization coils, liquid embolic systems, flow diversion stents, aneurysm clips, and others. Embolization coils refer to small, flexible devices made of materials such as platinum or stainless steel, used to obstruct blood flow in a vessel. It involves various treatments such as ischemic stroke solution, and hemorrhagic stroke solutions. It is provided for indications including brain aneurysms, arteriovenous malformation, and others. The end-users using this device are hospitals, ambulatory surgical centers, and others.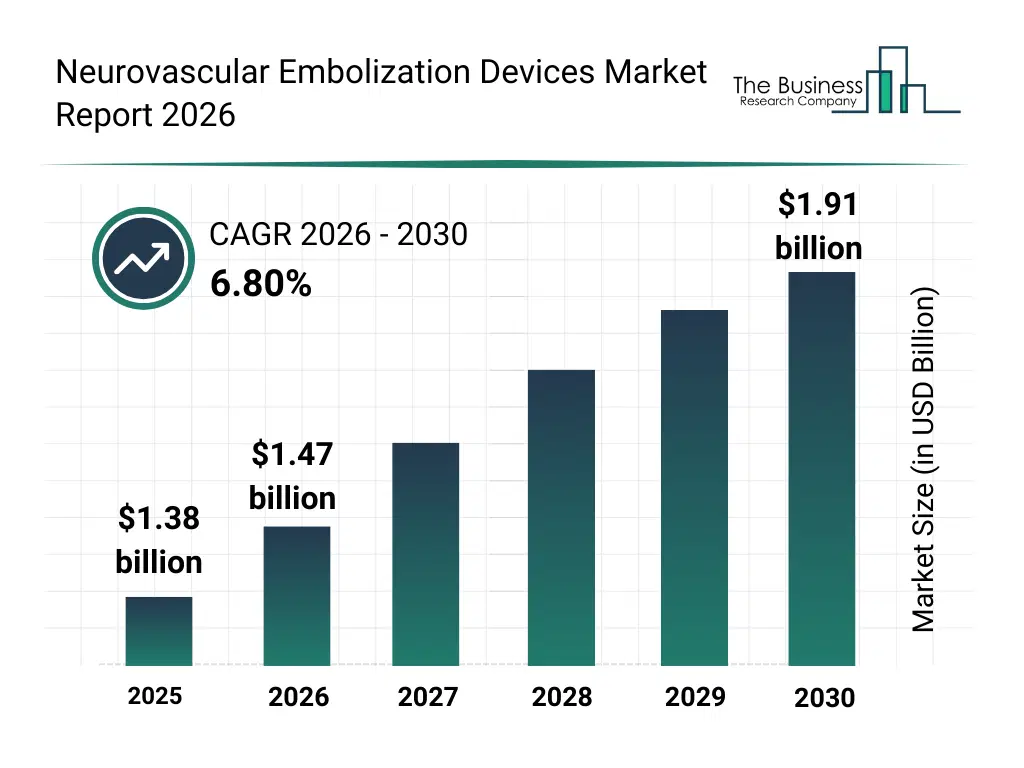
What Is The Neurovascular Embolization Devices Market Size and Share 2026?
The neurovascular embolization devices market size has grown strongly in recent years. It will grow from $1.38 billion in 2025 to $1.47 billion in 2026 at a compound annual growth rate (CAGR) of 6.2%. The growth in the historic period can be attributed to rising prevalence of cerebral aneurysms, expansion of neurointerventional procedures, increasing availability of skilled neurovascular specialists, growing hospital investments in advanced neurocare, wider adoption of minimally invasive stroke treatments.What Is The Neurovascular Embolization Devices Market Growth Forecast?
The neurovascular embolization devices market size is expected to see strong growth in the next few years. It will grow to $1.91 billion in 2030 at a compound annual growth rate (CAGR) of 6.8%. The growth in the forecast period can be attributed to increasing demand for advanced stroke management solutions, rising investments in neurovascular R&D, expansion of aging population with vascular disorders, growing adoption of next-generation embolization devices, increasing focus on improving procedural outcomes. Major trends in the forecast period include increasing adoption of flow diversion stents, rising use of liquid embolic systems, growing preference for minimally invasive neurointerventions, expansion of coil technologies with advanced coatings, enhanced focus on precision neurovascular devices.Global Neurovascular Embolization Devices Market Segmentation
1) By Product: Embolization Coils, Liquid Embolic Systems, Flow Diversion Stents, Aneurysm Clips, Other Products 2) By Treatment: Neurovascular Hemorrhage Management, Hemorrhagic Stroke Solutions 3) By Indication: Brain Aneurysms, Arteriovenous Malformation, Other Indications 4) By End User: Hospitals, Ambulatory Surgical Centers, Other End Users Subsegments: 1) By Embolization Coils: Detachable Coils, Bare Platinum Coils, Coils With Hydrogel Coatings 2) By Liquid Embolic Systems: N-Butyl Cyanoacrylate-Based Embolics, Onyx Liquid Embolic System 3) By Flow Diversion Stents: Stents With Dual-Layer Technology, Flow Diverters For Aneurysm Treatment 4) By Aneurysm Clips: Titanium Clips, Stainless Steel Clips 5) By Other Products: Balloons For Embolization, MicrocathetersWhat Is The Driver Of The Neurovascular Embolization Devices Market?
The rise in the prevalence of hemorrhages is expected to propel the neurovascular embolization devices market going forward. Hemorrhages are abnormal, uncontrolled bleeding that occurs internally within the body or externally through a break in the skin or mucous membrane. The prevalence of hemorrhages is due to several factors, such as aging populations, an increased incidence of hypertension, and the expanded use of anticoagulant medications. Neurovascular embolization devices are utilized in hemorrhagic strokes to obstruct abnormal blood vessels, reducing bleeding and preventing further damage to the brain. For instance, in February 2024, according to the research of King’s College London, a UK-based public research university, the number of cases and deaths from intracerebral hemorrhage (ICH) (a specific type of hemorrhage) is expected to increase by 59.4% in people aged 70 to 95 across the UK by 2050. Therefore, the rise in the prevalence of hemorrhages drives the growth of the neurovascular embolization devices industry.Key Players In The Global Neurovascular Embolization Devices Market
Major companies operating in the neurovascular embolization devices market are Johnson & Johnson, Abbott Laboratories, Medtronic plc, Stryker Corporation, Boston Scientific Corporation, Kaneka Corporation, Terumo Corporation, B Braun Melsungen AG, Cook Medical LLC, Integer Holdings Corporation, Merit Medical Systems Inc, Penumbra Inc, Asahi Intecc Co Ltd, MicroVention Inc, Balt USA LLC, Imperative Care Inc, Acandis GmbH & Co KG, Rapid Medical Ltd, Vesalio LLC, Phenox GmbH, Cerenovus, Sequent Medical IncGlobal Neurovascular Embolization Devices Market Trends and Insights
Major companies operating in the neurovascular embolization devices market are focusing on developing advanced solutions, such as next-generation flow diverters, to replace traditional aneurysm treatment methods. A next-generation flow diverter refers to a device that improves procedural safety and efficacy by enhancing vessel wall apposition, reducing clotting risk, and allowing for easier deployment in complex aneurysm anatomies. For instance, in July 2025, Stryker Corporation, a US-based medical technology company, launched the Surpass Elite Flow Diverter into its neurovascular portfolio. This innovation replaces older aneurysm treatment devices by providing minimally invasive flow diversion therapy for large or giant unruptured intracranial aneurysms. It delivers improved thrombo-resistance and optimized braid design while allowing physicians to achieve better procedural outcomes with less manipulation. Integrated with Stryker’s training and physician support programs, it offers a seamless clinical experience. The Surpass Elite Flow Diverter improves patient safety, simplifies procedural workflow, and helps expand adoption of flow diversion therapy in neurovascular care.What Are Latest Mergers And Acquisitions In The Neurovascular Embolization Devices Market?
In May 2023, Stryker Corporation, a US-based medical technology company, acquired Cerus Endovascular Ltd for $530 million. With this acquisition, Stryker aimed to enhance its capabilities in providing innovative medical devices for treating intracranial aneurysms, such as the contour neurovascular system and the neqstent coil-assisted flow diverter. Cerus Endovascular Ltd is an Ireland-based medical device company developing novel neurovascular embolization devices to treat intracranial aneurysms.Regional Outlook
North America was the largest region in the neurovascular embolization devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, SpainWhat Defines the Neurovascular Embolization Devices Market?
The neurovascular embolization devices market consists of sales of detachable balloons, particle embolic, vascular plugs, and embolization microspheres. Values in this market are ‘factory gate’ values, that is the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.Neurovascular Embolization Devices Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $1.47 billion |
| Revenue Forecast In 2035 | $1.91 billion |
| Growth Rate | CAGR of 6.2% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Product, Treatment, Indication, End User |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Johnson & Johnson, Abbott Laboratories, Medtronic plc, Stryker Corporation, Boston Scientific Corporation, Kaneka Corporation, Terumo Corporation, B Braun Melsungen AG, Cook Medical LLC, Integer Holdings Corporation, Merit Medical Systems Inc, Penumbra Inc, Asahi Intecc Co Ltd, MicroVention Inc, Balt USA LLC, Imperative Care Inc, Acandis GmbH & Co KG, Rapid Medical Ltd, Vesalio LLC, Phenox GmbH, Cerenovus, Sequent Medical Inc |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
Frequently Asked Questions
The Neurovascular Embolization Devices Market Report 2026 market was valued at $1.38 billion in 2025, increased to $1.47 billion in 2026, and is projected to reach $1.91 billion by 2030.
request a sample hereThe expected CAGR for the Neurovascular Embolization Devices Market Report 2026 market during the forecast period 2025–2030 is 6.8%.
request a sample hereMajor growth driver of the market includes: Rising Hemorrhages Drive Neurovascular Embolization Devices Market Growth in the Neurovascular Embolization Devices Market Report 2026 market. For further insights on this market,
request a sample hereThe neurovascular embolization devices market covered in this report is segmented –
1) By Product: Embolization Coils, Liquid Embolic Systems, Flow Diversion Stents, Aneurysm Clips, Other Products
2) By Treatment: Neurovascular Hemorrhage Management, Hemorrhagic Stroke Solutions
3) By Indication: Brain Aneurysms, Arteriovenous Malformation, Other Indications
4) By End User: Hospitals, Ambulatory Surgical Centers, Other End Users Subsegments:
1) By Embolization Coils: Detachable Coils, Bare Platinum Coils, Coils With Hydrogel Coatings
2) By Liquid Embolic Systems: N-Butyl Cyanoacrylate-Based Embolics, Onyx Liquid Embolic System
3) By Flow Diversion Stents: Stents With Dual-Layer Technology, Flow Diverters For Aneurysm Treatment
4) By Aneurysm Clips: Titanium Clips, Stainless Steel Clips
5) By Other Products: Balloons For Embolization, Microcatheters
request a sample here1) By Product: Embolization Coils, Liquid Embolic Systems, Flow Diversion Stents, Aneurysm Clips, Other Products
2) By Treatment: Neurovascular Hemorrhage Management, Hemorrhagic Stroke Solutions
3) By Indication: Brain Aneurysms, Arteriovenous Malformation, Other Indications
4) By End User: Hospitals, Ambulatory Surgical Centers, Other End Users Subsegments:
1) By Embolization Coils: Detachable Coils, Bare Platinum Coils, Coils With Hydrogel Coatings
2) By Liquid Embolic Systems: N-Butyl Cyanoacrylate-Based Embolics, Onyx Liquid Embolic System
3) By Flow Diversion Stents: Stents With Dual-Layer Technology, Flow Diverters For Aneurysm Treatment
4) By Aneurysm Clips: Titanium Clips, Stainless Steel Clips
5) By Other Products: Balloons For Embolization, Microcatheters
Major trend in this market includes: Stryker Introduces Innovative Flow Diverter for Enhanced Aneurysm Treatment For further insights on this market,
request a sample hereMajor companies operating in the Neurovascular Embolization Devices Market Report 2026 market are Major companies operating in the neurovascular embolization devices market are Johnson & Johnson, Abbott Laboratories, Medtronic plc, Stryker Corporation, Boston Scientific Corporation, Kaneka Corporation, Terumo Corporation, B Braun Melsungen AG, Cook Medical LLC, Integer Holdings Corporation, Merit Medical Systems Inc, Penumbra Inc, Asahi Intecc Co Ltd, MicroVention Inc, Balt USA LLC, Imperative Care Inc, Acandis GmbH & Co KG, Rapid Medical Ltd, Vesalio LLC, Phenox GmbH, Cerenovus, Sequent Medical Inc
request a sample hereNorth America was the largest region in the neurovascular embolization devices market in 2025. Asia-Pacific is expected to be the fastest-growing region in the forecast period. The regions covered in the neurovascular embolization devices market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa.
request a sample here