
Typhoid Vi Polysaccharide Vaccine Market Report 2026
Global Outlook – By Type (Child, Adult ), By Route Of Administration (Oral, Parenteral ), By Application (Government Institution, Private Sector, Other Applications ) - Market Size, Trends, And Global Forecast 2026-2035
Typhoid Vi Polysaccharide Vaccine Market Overview
• Typhoid Vi Polysaccharide Vaccine market size has reached to $6.71 billion in 2025 • Expected to grow to $15.51 billion in 2030 at a compound annual growth rate (CAGR) of 18.3% • Growth Driver: Escalating Typhoid Prevalence Fuels Expansion In The Typhoid Vi Polysaccharide Vaccine Market • Market Trend: Innovative Collaborations And Strategic Investments In Typhoid Vi Polysaccharide Vaccine Development • Asia-Pacific was the largest region in 2025.What Is Covered Under Typhoid Vi Polysaccharide Vaccine Market?
Typhoid VI polysaccharide vaccine refers to the recommendation of active immunization for people aged two and up, concerning the prevention of typhoid fever caused by Salmonella typhi. The main types of typhoid VI polysaccharide vaccine are child and adult. A child is a person between birth and puberty, or full growth. The routes of administration for this vaccine include oral and parenteral and are applied in government institutions, the private sector, and others.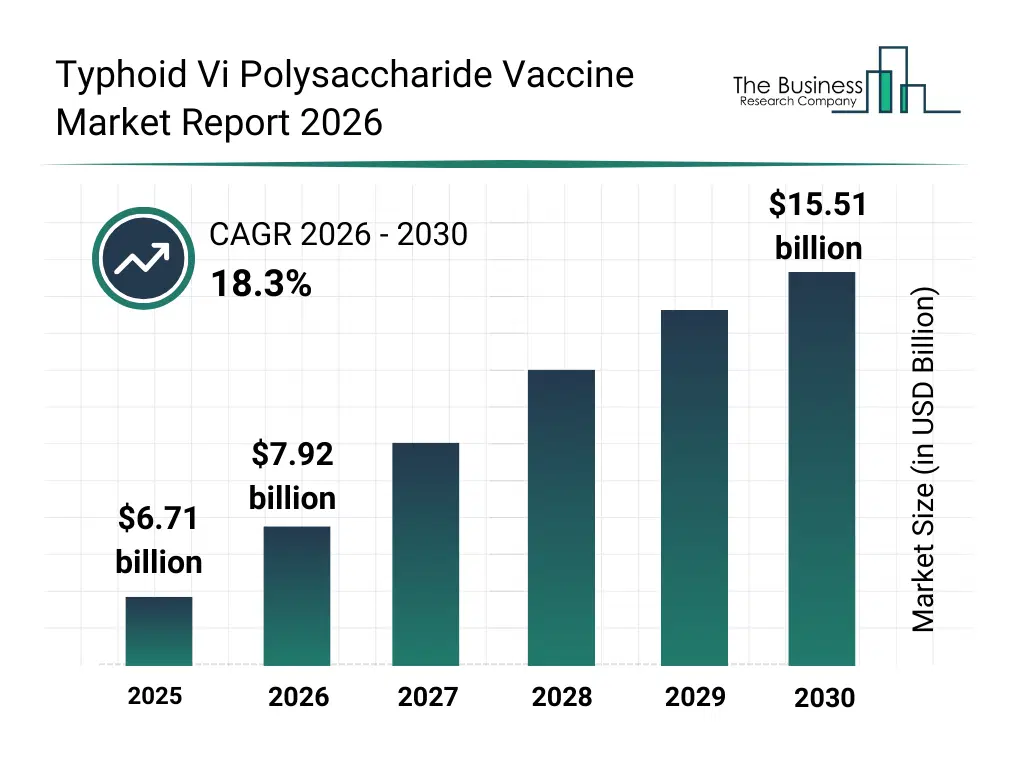
What Is The Typhoid Vi Polysaccharide Vaccine Market Size and Share 2026?
The typhoid vi polysaccharide vaccine market size has grown rapidly in recent years. It will grow from $6.71 billion in 2025 to $7.92 billion in 2026 at a compound annual growth rate (CAGR) of 18.1%. The growth in the historic period can be attributed to expansion of national immunization schedules, increased awareness of typhoid prevention, availability of polysaccharide vaccine formulations, growth of public vaccination campaigns, support from global health organizations.What Is The Typhoid Vi Polysaccharide Vaccine Market Growth Forecast?
The typhoid vi polysaccharide vaccine market size is expected to see rapid growth in the next few years. It will grow to $15.51 billion in 2030 at a compound annual growth rate (CAGR) of 18.3%. The growth in the forecast period can be attributed to increasing focus on mass immunization in endemic regions, rising investments in vaccine manufacturing capacity, expansion of adult vaccination programs, growing emphasis on travel-related immunization, increasing adoption of improved vaccine storage technologies. Major trends in the forecast period include increasing government-led immunization programs, rising demand for pediatric vaccination coverage, growing focus on preventive healthcare measures, expansion of public-private vaccine distribution models, enhanced emphasis on cold chain optimization.Global Typhoid Vi Polysaccharide Vaccine Market Segmentation
1) By Type: Child, Adult 2) By Route Of Administration: Oral, Parenteral 3) By Application: Government Institution, Private Sector, Other Applications Subsegments: 1) By Child: Pediatric Formulations, Age-Specific Dosages 2) By Adult: Standard Adult Formulations, High-Risk Population FormulationsWhat Are The Drivers Of The Typhoid Vi Polysaccharide Vaccine Market?
Rising typhoid prevalence is expected to propel the growth of the typhoid Vi polysaccharide vaccine market going forward. Typhoid is an infection caused by the bacteria Salmonella Typhi that can be fatal and typically spreads through contaminated food or drink. The rise in typhoid prevalence is driven by poor sanitation, unsafe water sources, rapid urbanization, and increased exposure to contaminated food and water. The typhoid Vi polysaccharide vaccine market supports this rise by providing immunization for adults and children aged 2 years and older, offering protective immunity against Salmonella enterica serovar Typhi and reducing the burden of typhoid fever in endemic regions. For instance, in August 2024, according to data published by the UK Health Security Agency, laboratory-confirmed symptomatic enteric fever cases in England, Wales, and Northern Ireland increased with S. Typhi reaching 313 cases, S. Paratyphi A reaching 135 cases, and S. Paratyphi B reaching 18 cases. Therefore, the rising typhoid prevalence is driving the growth of the typhoid Vi polysaccharide vaccine industry. The growing immunization campaigns are expected to propel the growth of the typhoid Vi polysaccharide vaccine market going forward. Immunization campaigns refer to delivery strategies used to quickly reach large numbers of individuals with one or more vaccines at national or sub-national levels. The rise in immunization campaigns is driven by increasing public-health focus on typhoid prevention in high-burden regions and the need to reduce antimicrobial resistance through widespread vaccination. Growing immunization campaigns support the Typhoid Vi polysaccharide vaccine by increasing vaccine awareness, accessibility, and coverage, ultimately reducing the incidence and transmission of typhoid fever. For instance, in June 2023, according to the UK Health Security Agency, a UK-based government agency, total of 3,502,566 children from Reception to Year 9 were vaccinated as part of the school-aged vaccination program in 2022–2023. Therefore, growing immunization campaigns are driving the growth of the typhoid Vi polysaccharide vaccine industry.Key Players In The Global Typhoid Vi Polysaccharide Vaccine Market
Major companies operating in the typhoid vi polysaccharide vaccine market are Sanofi S.A., Merck & Co. Inc., PT Bio Farma, Bharat Biotech International Limited, Bio-Med Pvt. Ltd., Hualan Biological Engineering Inc., Indian Immunologicals Limited, Serum Institute of India Limited, Biological E Limited, Panacea Biotec Limited, Zydus Lifesciences Limited, Beijing Minhai Biotechnology Co. Ltd., Lanzhou Institute of Biological Products Co. Ltd., Chengdu Institute of Biological Products Co. Ltd., Wuhan Institute of Biological Products Co. Ltd., Eubiologics Co. Ltd., Incepta Vaccine Limited, GC Pharma, KM Biologics Inc., Bharat Immunologicals and Biologicals Corporation Limited, Sinovac Biotech Ltd., Bio Farma IndonesiaGlobal Typhoid Vi Polysaccharide Vaccine Market Trends and Insights
Major companies operating in the typhoid Vi polysaccharide vaccine market are focusing on strengthening their market position through strategic investments, such as funding partnerships for next-generation vaccine delivery platforms, to accelerate innovation and expand global immunization reach. Funding partnerships for next-generation vaccine delivery platforms refer to collaborative investment initiatives aimed at developing improved typhoid vaccination technologies particularly needle-free, thermostable, and easy-to-administer formats that enhance accessibility, coverage, and compliance in low-resource settings. For instance, in August 2023, SK Bioscience, a South Korea-based biotech company, together with Vaxxas, an Australia-based biotechnology company specializing in needle-free vaccine delivery, secured AUD 0.0054 billion (USD 0.00367 billion) in funding from Wellcome, a UK-based charitable foundation, to develop a typhoid conjugate vaccine delivered via a needle-free high-density microarray patch (HD-MAP). This innovation features a micro-projection patch that eliminates the need for traditional needles, thermostability that supports distribution in low-infrastructure regions, and the potential for simplified, pain-free administration, targeting improved global uptake of typhoid vaccination.Regional Outlook
Asia-Pacific was the largest region in the typhoid Vi polysaccharide vaccine market share in 2025. The regions covered in this market report are Asia-Pacific, South East Asia, Western Europe, Eastern Europe, North America, South America, Middle East, Africa. The countries covered in this market report are Australia, Brazil, China, France, Germany, India, Indonesia, Japan, Taiwan, Russia, South Korea, UK, USA, Canada, Italy, Spain.What Defines the Typhoid Vi Polysaccharide Vaccine Market?
The typhoid Vi polysaccharide vaccine market consists of sales of Vi capsular polysaccharide vaccine, and non-conjugated Vi polysaccharide vaccine. Values in this market are ‘factory gate’ values, that is, the value of goods sold by the manufacturers or creators of the goods, whether to other entities (including downstream manufacturers, wholesalers, distributors, and retailers) or directly to end customers. The value of goods in this market includes related services sold by the creators of the goods.How is Market Value Defined and Measured?
The market value is defined as the revenues that enterprises gain from the sale of goods and/or services within the specified market and geography through sales, grants, or donations in terms of the currency (in USD unless otherwise specified). The revenues for a specified geography are consumption values that are revenues generated by organizations in the specified geography within the market, irrespective of where they are produced. It does not include revenues from resales along the supply chain, either further along the supply chain or as part of other products.What Key Data and Analysis Are Included in the Typhoid Vi Polysaccharide Vaccine Market Report 2026?
The typhoid vi polysaccharide vaccine market research report is one of a series of new reports from The Business Research Company that provides market statistics, including industry global market size, regional shares, competitors with the market share, detailed market segments, market trends and opportunities, and any further data you may need to thrive in the typhoid vi polysaccharide vaccine industry. The market research report delivers a complete perspective of everything you need, with an in-depth analysis of the current and future state of the industry.Typhoid Vi Polysaccharide Vaccine Market Report Forecast Analysis
| Report Attribute | Details |
|---|---|
| Market Size Value In 2026 | $7.92 billion |
| Revenue Forecast In 2035 | $15.51 billion |
| Growth Rate | CAGR of 18.1% from 2026 to 2035 |
| Base Year For Estimation | 2025 |
| Actual Estimates/Historical Data | 2020-2025 |
| Forecast Period | 2026 - 2030 - 2035 |
| Market Representation | Revenue in USD Billion and CAGR from 2026 to 2035 |
| Segments Covered | Type, Route Of Administration, Application |
| Regional Scope | Asia-Pacific, Western Europe, Eastern Europe, North America, South America, Middle East, Africa |
| Country Scope | The countries covered in the report are Australia, Brazil, China, France, Germany, India, ... |
| Key Companies Profiled | Sanofi S.A., Merck & Co. Inc., PT Bio Farma, Bharat Biotech International Limited, Bio-Med Pvt. Ltd., Hualan Biological Engineering Inc., Indian Immunologicals Limited, Serum Institute of India Limited, Biological E Limited, Panacea Biotec Limited, Zydus Lifesciences Limited, Beijing Minhai Biotechnology Co. Ltd., Lanzhou Institute of Biological Products Co. Ltd., Chengdu Institute of Biological Products Co. Ltd., Wuhan Institute of Biological Products Co. Ltd., Eubiologics Co. Ltd., Incepta Vaccine Limited, GC Pharma, KM Biologics Inc., Bharat Immunologicals and Biologicals Corporation Limited, Sinovac Biotech Ltd., Bio Farma Indonesia |
| Customization Scope | Request for Customization |
| Pricing And Purchase Options | Explore Purchase Options |
